Epstein-Barr virus-encoded LMP1 regulated Pim1 kinase expression promotes nasopharyngeal carcinoma cells proliferation
- PMID: 30809095
- PMCID: PMC6376889
- DOI: 10.2147/OTT.S190274
Epstein-Barr virus-encoded LMP1 regulated Pim1 kinase expression promotes nasopharyngeal carcinoma cells proliferation
Abstract
Background: Epstein-Barr virus-encoded LMP1 plays a critical role in the carcinogenesis of nasopharyngeal carcinoma (NPC), but the mechanism remains elusive. We aimed to analyze the expression and clinical pathological significance of provirus integration site for Moloney murine leukemia virus 1 (Pim1) in clinical NPC, and to elucidate the effect of LMP1 on Pim1 expression and its mechanism.
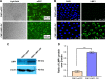
Methods: Immunohistochemical staining was used to detect the expression of Pim1 in clinical NPC tissues and control nasopharyngeal chronic inflammation (NPI) tissues, and the correlation between Pim1 and clinical parameters of NPC patients was analyzed. The LMP1 stable expression cell line CNE1-LMP1-OV was constructed through infecting the well-differentiated nasopharyngeal carcinoma cells CNE1 with LMP1 overexpressing lentivirus. Then the in vivo experiments were conducted.
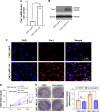
Results: Among 89 NPC patients, 48 cases (53.93%) were positive for Pim1, while only one case was Pim1 positive in 15 NPI controls (6.67%). Pim1 expression was not correlated with gender, age, smoking status and clinical classification of NPC patients, but positively correlated with T, N and M classification. CNE1-LMP1-OV cell line was successfully established, which displayed a higher cell proliferation ability and Pim1 expression. NF-κB inhibitor PDTC, PKC inhibitor GF109203X and STAT3 inhibitor Stattic significantly attenuated LMP1-induced Pim1 expression, and while AP-1 inhibitor SR11302 showed no inhibitory effect. Interestingly, Pim1 inhibitor quercetagetin significantly inhibited the proliferation of CNE1-LMP1-OV cells.
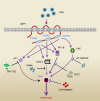
Conclusion: LMP1 mediates Pim1 expression through NF-κB, PKC and STAT3 signaling, which promotes the proliferation of NPC cells and participate in the clinical progression of NPC.
Keywords: LMP1; Pim1; cell proliferation; nasopharyngeal carcinoma.
Conflict of interest statement
Disclosure The authors report no conflicts of interest in this work.
Figures



References
-
- Merkel AL, Meggers E, Ocker M. PIM1 kinase as a target for cancer therapy. Expert Opin Investig Drugs. 2012;21(4):425–436. - PubMed
LinkOut - more resources
Full Text Sources
Miscellaneous

