The Brain NGF Metabolic Pathway in Health and in Alzheimer's Pathology
- PMID: 30809111
- PMCID: PMC6379336
- DOI: 10.3389/fnins.2019.00062
The Brain NGF Metabolic Pathway in Health and in Alzheimer's Pathology
Abstract
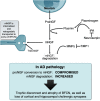
Emerging research has re-emphasized the role of the cortical cholinergic system in the symptomology and progression of Alzheimer's disease (AD). Basal forebrain (BF) cholinergic nuclei depend on target-derived NGF for survival during development and for the maintenance of a classical cholinergic phenotype during adulthood. In AD, BF cholinergic neurons lose their cholinergic phenotype and function, suggesting an impairment in NGF-mediated trophic support. We propose that alterations to the enzymatic pathway that controls the maturation of proNGF to mature NGF and the latter's ulterior degradation underlie this pathological process. Indeed, the NGF metabolic pathway has been demonstrated to be impaired in AD and other amyloid pathologies, and pharmacological manipulation of NGF metabolism has consequences in vivo for both levels of proNGF/NGF and the phenotype of BF cholinergic neurons. The NGF pathway may also have potential as a biomarker of cognitive decline in AD, as its changes can predict future cognitive decline in patients with Down syndrome as they develop preclinical Alzheimer's pathology. New evidence suggests that the cholinergic system, and by extension NGF, may have a greater role in the progression of AD than previously realized, as changes to the BF precede and predict changes to the entorhinal cortex, as anticholinergic drugs increase odds of developing AD, and as the use of donepezil can reduce rates of hippocampal and cortical thinning. These findings suggest that new, more sophisticated cholinergic therapies should be capable of preserving the basal forebrain thus having profound positive effects as treatments for AD.
Keywords: Alzheimer’s disease; basal forebrain cholinergic nuclei; cholinergic system; nerve growth factor; trophic support.
Figures
References
Publication types
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous