Modulation of microbial communities and mucosal gene expression in chicken intestines after galactooligosaccharides delivery In Ovo
- PMID: 30811518
- PMCID: PMC6392319
- DOI: 10.1371/journal.pone.0212318
Modulation of microbial communities and mucosal gene expression in chicken intestines after galactooligosaccharides delivery In Ovo
Abstract
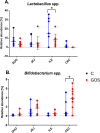
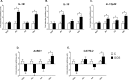
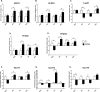
Intestinal mucosa is the interface between the microbial content of the gut and the host's milieu. The goal of this study was to modulate chicken intestinal microflora by in ovo stimulation with galactooligosaccharides (GOS) prebiotic and to demonstrate the molecular responses of the host. The animal trial was performed on meat-type chickens (Ross 308). GOS was delivered by in ovo injection performed into the air cell on day 12 of egg incubation. Analysis of microbial communities and mucosal gene expression was performed at slaughter (day 42 post-hatching). Chyme (for DNA isolation) and intestinal mucosa (for RNA isolation) from four distinct intestinal segments (duodenum, jejunum, ileum, and caecum) was sampled. The relative abundance of Bifidobacterium spp. and Lactobacillus spp. in DNA isolated from chyme samples was determined using qPCR. On the host side, the mRNA expression of 13 genes grouped into two panels was analysed with RT-qPCR. Panel (1) included genes related to intestinal innate immune responses (IL-1β, IL-10 and IL-12p40, AvBD1 and CATHL2). Panel (2) contained genes involved in intestinal barrier function (MUC6, CLDN1 and TJAP1) and nutrients sensing (FFAR2 and FFAR4, GLUT1, GLUT2 and GLUT5). GOS increased the relative abundance of Bifidobacterium in caecum (from 1.3% to 3.9%). Distinct effects of GOS on gene expression were manifested in jejunum and caecum. Cytokine genes (IL-1β, IL-10 and IL-12p40) were up-regulated in the jejunum and caecum of the GOS-treated group. Host defence peptides (AvBD1 and CATHL2) were up-regulated in the caecum of the GOS-treated group. Free fatty acid receptors (FFAR2 and FFAR4) were up-regulated in all three compartments of the intestine (except the duodenum). Glucose transporters were down-regulated in duodenum (GLUT2 and GLUT5) but up-regulated in the hindgut (GLUT1 and GLUT2). In conclusion, GOS delivered in ovo had a bifidogenic effect in adult chickens. It also modulated gene expression related to intestinal immune responses, gut barrier function, and nutrient sensing.
Conflict of interest statement
The authors have declared that no competing interests exist.
Figures

Similar articles
-
Impact of in ovo administration of xylo- and mannooligosaccharides on broiler chicken gut health.Poult Sci. 2024 Dec;103(12):104261. doi: 10.1016/j.psj.2024.104261. Epub 2024 Aug 24. Poult Sci. 2024. PMID: 39265513 Free PMC article.
-
Transcriptome modulation by in ovo delivered Lactobacillus synbiotics in a range of chicken tissues.Gene. 2019 May 25;698:27-33. doi: 10.1016/j.gene.2019.02.068. Epub 2019 Mar 1. Gene. 2019. PMID: 30831211
-
Avian model to mitigate gut-derived immune response and oxidative stress during heat.Biosystems. 2019 Apr;178:10-15. doi: 10.1016/j.biosystems.2019.01.007. Epub 2019 Jan 16. Biosystems. 2019. PMID: 30659866
-
Prebiotics and synbiotics - in ovo delivery for improved lifespan condition in chicken.BMC Vet Res. 2018 Dec 17;14(1):402. doi: 10.1186/s12917-018-1738-z. BMC Vet Res. 2018. PMID: 30558599 Free PMC article. Review.
-
Factors influencing the development of gastrointestinal tract and nutrient transporters' function during the embryonic life of chickens-A review.J Anim Physiol Anim Nutr (Berl). 2023 Nov;107(6):1419-1428. doi: 10.1111/jpn.13852. Epub 2023 Jul 6. J Anim Physiol Anim Nutr (Berl). 2023. PMID: 37409520 Review.
Cited by
-
Novel strategies to improve chicken performance and welfare by unveiling host-microbiota interactions through hologenomics.Front Physiol. 2022 Sep 6;13:884925. doi: 10.3389/fphys.2022.884925. eCollection 2022. Front Physiol. 2022. PMID: 36148301 Free PMC article.
-
In ovo supplementation of chitooligosaccharide and chlorella polysaccharide affects cecal microbial community, metabolic pathways, and fermentation metabolites in broiler chickens.Poult Sci. 2020 Oct;99(10):4776-4785. doi: 10.1016/j.psj.2020.06.061. Epub 2020 Jul 22. Poult Sci. 2020. PMID: 32988512 Free PMC article.
-
Modulation of Intestinal Histology by Probiotics, Prebiotics and Synbiotics Delivered In Ovo in Distinct Chicken Genotypes.Animals (Basel). 2021 Nov 18;11(11):3293. doi: 10.3390/ani11113293. Animals (Basel). 2021. PMID: 34828024 Free PMC article.
-
Free Fatty Acid Receptors 2 and 3 as Microbial Metabolite Sensors to Shape Host Health: Pharmacophysiological View.Biomedicines. 2020 Jun 8;8(6):154. doi: 10.3390/biomedicines8060154. Biomedicines. 2020. PMID: 32521775 Free PMC article. Review.
-
Inter- and Transgenerational Effects of In Ovo Stimulation with Bioactive Compounds on Cecal Tonsils and Cecal Mucosa Transcriptomes in a Chicken Model.Int J Mol Sci. 2025 Jan 29;26(3):1174. doi: 10.3390/ijms26031174. Int J Mol Sci. 2025. PMID: 39940944 Free PMC article.
References
-
- Duke G. Alimentary canal: anatomy, regulation of feeding, and motility Avian physiology: Springer; 1986. p. 269–88.
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Miscellaneous

