Extreme Fuzziness: Direct Interactions between Two IDPs
- PMID: 30813629
- PMCID: PMC6468500
- DOI: 10.3390/biom9030081
Extreme Fuzziness: Direct Interactions between Two IDPs
Abstract
Protein interactions involving intrinsically disordered proteins (IDPs) greatly extend the range of binding mechanisms available to proteins. In interactions employing coupled folding and binding, IDPs undergo disorder-to-order transitions to form a complex with a well-defined structure. In many other cases, IDPs retain structural plasticity in the final complexes, which have been defined as the fuzzy complexes. While a large number of fuzzy complexes have been characterized with variety of fuzzy patterns, many of the interactions are between an IDP and a structured protein. Thus, whether two IDPs can interact directly to form a fuzzy complex without disorder-to-order transition remains an open question. Recently, two studies of interactions between IDPs (4.1G-CTD/NuMA and H1/ProTα) have found a definite answer to this question. Detailed characterizations combined with nuclear magnetic resonance (NMR), single-molecule Förster resonance energy transfer (smFRET) and molecular dynamics (MD) simulation demonstrate that direct interactions between these two pairs of IDPs do form fuzzy complexes while retaining the conformational dynamics of the isolated proteins, which we name as the extremely fuzzy complexes. Extreme fuzziness completes the full spectrum of protein-protein interaction modes, suggesting that a more generalized model beyond existing binding mechanisms is required. Previous models of protein interaction could be applicable to some aspects of the extremely fuzzy interactions, but in more general sense, the distinction between native and nonnative contacts, which was used to understand protein folding and binding, becomes obscure. Exploring the phenomenon of extreme fuzziness may shed new light on molecular recognition and drug design.
Keywords: binding mechanism; extremely fuzzy complex; intrinsic disordered protein; protein interaction.
Conflict of interest statement
The authors declare no conflict of interest.
Figures
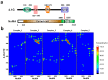
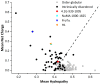
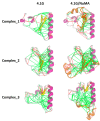
References
-
- Linderstrøm-Lang K.U., Schellman J.A. Protein Structure and Enzyme Activity. Enzyme. 1959;1:443–510.
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

