Intramuscular 17-hydroxyprogesterone caproate to prevent preterm birth among HIV-infected women in Zambia: study protocol of the IPOP randomized trial
- PMID: 30813934
- PMCID: PMC6391830
- DOI: 10.1186/s12884-019-2224-8
Intramuscular 17-hydroxyprogesterone caproate to prevent preterm birth among HIV-infected women in Zambia: study protocol of the IPOP randomized trial
Abstract
Background: Each year, an estimated 15 million babies are born preterm, a global burden borne disproportionately by families in lower-income countries. Maternal HIV infection increases a woman's risk of delivering prematurely, and antiretroviral therapy (ART) may compound this risk. While prenatal progesterone prophylaxis prevents preterm birth among some high-risk women, it is unknown whether HIV-infected women could benefit from this therapy. We are studying the efficacy of progesterone supplementation to reduce the risk of preterm birth among pregnant women with HIV in Lusaka, Zambia.
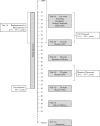
Methods: The Improving Pregnancy Outcomes with Progesterone (IPOP) study is a Phase III double-masked, placebo-controlled, randomized trial of intramuscular 17-alpha hydroxprogesterone caproate (17P) to prevent preterm birth in HIV-infected women. A total of 800 women will be recruited prior to 24 weeks of gestation and randomly allocated to 17P or placebo administered by weekly intramuscular injection. The primary outcome will be a composite of live birth prior to 37 completed gestational weeks or stillbirth at any gestational age. Secondary outcomes will include very preterm birth (< 34 weeks), extreme preterm birth (< 28 weeks), small for gestational age (<10th centile), low birth weight (< 2500 g), and neonatal outcomes. In secondary analysis, we will assess whether specific HIV-related covariates, including the timing of maternal ART initiation relative to conception, is associated with progesterone's prophylactic efficacy, if any.
Discussion: We hypothesize that weekly prenatal 17P will reduce the risk of HIV-related preterm birth. An inexpensive intervention to prevent preterm birth among pregnant women with HIV could have substantial global public health impact.
Trial registration: NCT03297216 ; September 29, 2017.
Keywords: 17-alpha hydroxyprogesterone caproate; Antiretroviral therapy; HIV; Preterm birth; Progesterone; Sub-Saharan Africa.
Conflict of interest statement
Ethics approval and consent to participate
This study was approved by the University of North Carolina Institutional Review Board, reference #17–1173, and the University of Zambia Biomedical Research Ethics Committee, reference #015–06-17, and the Zambia Medicines Regulatory Authority, reference CT-070. Written informed consent is obtained from all participants prior to study enrollment.
Consent for publication
Not applicable
Competing interests
The authors declare that they have no competing interests.
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Figures
References
-
- March of Dimes P, Save the Children, WHO. Born too Soon: the Global Action Report on Preterm Birth. Geneva: WHO;2012.
-
- Blencowe H, Cousens S, Oestergaard MZ, et al. National, regional, and worldwide estimates of preterm birth rates in the year 2010 with time trends since 1990 for selected countries: a systematic analysis and implications. Lancet. 2012;379(9832):2162–2172. doi: 10.1016/S0140-6736(12)60820-4. - DOI - PubMed
-
- Joint United Nations Programme on HIV/AIDS. The Gap Report. 2014; http://www.unaids.org/sites/default/files/media_asset/UNAIDS_Gap_report_.... Accessed 26 Jan 2016.
Publication types
MeSH terms
Substances
Associated data
Grants and funding
LinkOut - more resources
Full Text Sources
Medical