Generation of microalga Chlamydomonas reinhardtii expressing shrimp antiviral dsRNA without supplementation of antibiotics
- PMID: 30816201
- PMCID: PMC6395707
- DOI: 10.1038/s41598-019-39539-x
Generation of microalga Chlamydomonas reinhardtii expressing shrimp antiviral dsRNA without supplementation of antibiotics
Abstract
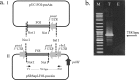
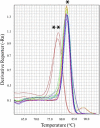
RNA interference (RNAi) is an effective way of combating shrimp viruses by using sequence-specific double-stranded (dsRNA) designed to knock down key viral genes. The aim of this study was to use microalgae expressing antiviral dsRNA as a sustainable feed supplement for shrimp offering viral protection. In this proof of concept, we engineered the chloroplast genome of the green microalga Chlamydomonas reinhardtii for the expression of a dsRNA cassette targeting a shrimp yellow head viral gene. We used a previously described chloroplast transformation approach that allows for the generation of stable, marker-free C. reinhardtii transformants without the supplementation of antibiotics. The generated dsRNA-expressing microalgal strain was then used in a shrimp feeding trial to evaluate the efficiency of the algal RNAi-based vaccine against the virus. Shrimps treated with dsRNA-expressed algal cells prior to YHV infection had 50% survival at 8 day-post infection (dpi), whereas 84.1% mortality was observed in control groups exposed to the YHV virus. RT-PCR using viral specific primers revealed a lower infection rate in dsRNA-expressing algae treated shrimp (55.6 ± 11.1%) compared to control groups (88.9 ± 11.1% and 100.0 ± 0.0%, respectively). Our results are promising for using microalgae as a novel, sustainable alternative as a nutritious, anti-viral protective feedstock in shrimp aquaculture.
Conflict of interest statement
The authors declare no competing interests.
Figures




References
-
- Enzing, C., Ploeg, M., Barbosa, M. & Sijtsma, L. Microalgae-based products for the food and feed sector: an outlook forEurope. JRC Scientific and policy reports 19–37 (2014).
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

