Phase IB/II Randomized Study of FOLFIRINOX Plus Pegylated Recombinant Human Hyaluronidase Versus FOLFIRINOX Alone in Patients With Metastatic Pancreatic Adenocarcinoma: SWOG S1313
- PMID: 30817250
- PMCID: PMC6494359
- DOI: 10.1200/JCO.18.01295
Phase IB/II Randomized Study of FOLFIRINOX Plus Pegylated Recombinant Human Hyaluronidase Versus FOLFIRINOX Alone in Patients With Metastatic Pancreatic Adenocarcinoma: SWOG S1313
Abstract
Purpose: Pegylated recombinant human hyaluronidase (PEGPH20) degrades hyaluronan (HA) and, in combination with chemotherapy, prolongs survival in preclinical models. The activity of PEGPH20 with modified fluorouracil, leucovorin, irinotecan, and oxaliplatin (mFOLFIRINOX) was evaluated in patients with metastatic pancreatic cancer (mPC).
Materials and methods: Patients had untreated mPC, a performance status of 0 to 1, and adequate organ function. Tumor HA status was not required for eligibility. After a phase Ib dose-finding study of mFOLFIRINOX plus PEGPH20, the phase II open-label study randomly assigned patients (1:1) to the combination arm or to mFOLFIRINOX alone (n = 138). The primary end point was overall survival (OS).
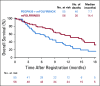
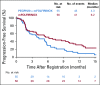
Results: PEGPH20 dosages of 3 µg/kg every 2 weeks were more tolerable than twice-weekly dosages used in the phase I study, so 3 µg/kg every 2 weeks was the phase II dosage. An amendment instituted enoxaparin prophylaxis in the PEGPH20 combination arm as a result of increased thromboembolic (TE) events. The planned interim futility analysis when 35 deaths (of 103 analyzable patients) occurred resulted in an OS hazard ratio (HR) of 2.07 that favored the control arm, and the study was closed to accrual. The treatment-related grade 3 to 4 toxicity was significantly increased in the PEGPH20 combination arm relative to control (odds ratio, 2.7; 95% CI, 1.1 to 7.1). The median OS in the mFOLFIRINOX arm was 14.4 months (95% CI, 10.1 to 15.7 months) versus 7.7 months (95% CI, 4.6 to 9.3 months) in the PEGPH20 combination arm.
Conclusion: Addition of PEGPH20 to mFOLFIRINOX seems to be detrimental in patients unselected for tumor HA status. This combination caused increased toxicity (mostly GI and TE events) and resulted in decreased treatment duration compared with mFOLFIRINOX alone. The median OS in the mFOLFIRINOX control arm (14.4 months) is, to our knowledge, the longest yet reported and can be considered for patients with good PS.
Trial registration: ClinicalTrials.gov NCT01959139.
Figures
Comment in
-
Targeting Stroma: A Tale of Caution.J Clin Oncol. 2019 May 1;37(13):1041-1043. doi: 10.1200/JCO.19.00056. Epub 2019 Mar 12. J Clin Oncol. 2019. PMID: 30860950 No abstract available.
References
-
- Conroy T, Desseigne F, Ychou M, et al. FOLFIRINOX versus gemcitabine for metastatic pancreatic cancer. N Engl J Med. 2011;364:1817–1825. - PubMed
Publication types
MeSH terms
Substances
Associated data
Grants and funding
- U10 CA180826/CA/NCI NIH HHS/United States
- UG1 CA189830/CA/NCI NIH HHS/United States
- U10 CA180828/CA/NCI NIH HHS/United States
- UG1 CA189853/CA/NCI NIH HHS/United States
- UG1 CA233163/CA/NCI NIH HHS/United States
- U10 CA046368/CA/NCI NIH HHS/United States
- U10 CA180888/CA/NCI NIH HHS/United States
- U10 CA180830/CA/NCI NIH HHS/United States
- UL1 TR001863/TR/NCATS NIH HHS/United States
- N01 CA013612/CA/NCI NIH HHS/United States
- U10 CA013612/CA/NCI NIH HHS/United States
- U10 CA180835/CA/NCI NIH HHS/United States
- UG1 CA189848/CA/NCI NIH HHS/United States
- U10 CA180846/CA/NCI NIH HHS/United States
- UG1 CA180830/CA/NCI NIH HHS/United States
- UG1 CA189858/CA/NCI NIH HHS/United States
- U10 CA180819/CA/NCI NIH HHS/United States
- UG1 CA189953/CA/NCI NIH HHS/United States
- UG1 CA233328/CA/NCI NIH HHS/United States
LinkOut - more resources
Full Text Sources
Medical

