Chloride Channels in Astrocytes: Structure, Roles in Brain Homeostasis and Implications in Disease
- PMID: 30818802
- PMCID: PMC6429410
- DOI: 10.3390/ijms20051034
Chloride Channels in Astrocytes: Structure, Roles in Brain Homeostasis and Implications in Disease
Abstract
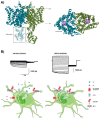
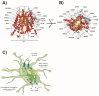
Astrocytes are the most abundant cell type in the CNS (central nervous system). They exert multiple functions during development and in the adult CNS that are essential for brain homeostasis. Both cation and anion channel activities have been identified in astrocytes and it is believed that they play key roles in astrocyte function. Whereas the proteins and the physiological roles assigned to cation channels are becoming very clear, the study of astrocytic chloride channels is in its early stages. In recent years, we have moved from the identification of chloride channel activities present in astrocyte primary culture to the identification of the proteins involved in these activities, the determination of their 3D structure and attempts to gain insights about their physiological role. Here, we review the recent findings related to the main chloride channels identified in astrocytes: the voltage-dependent ClC-2, the calcium-activated bestrophin, the volume-activated VRAC (volume-regulated anion channel) and the stress-activated Maxi-Cl-. We discuss key aspects of channel biophysics and structure with a focus on their role in glial physiology and human disease.
Keywords: astrocyte; chloride channel; human diseases; physiology; structure.
Conflict of interest statement
The authors declare no conflict of interest.
Figures


Similar articles
-
LRRC8A is essential for swelling-activated chloride current and for regulatory volume decrease in astrocytes.FASEB J. 2019 Jan;33(1):101-113. doi: 10.1096/fj.201701397RR. Epub 2018 Jun 29. FASEB J. 2019. PMID: 29957062
-
Expression of novel isoforms of the CIC-1 chloride channel in astrocytic glial cells in vitro.Glia. 2004 Jul;47(1):46-57. doi: 10.1002/glia.20024. Glia. 2004. PMID: 15139012
-
Pharmacological comparison of swelling-activated excitatory amino acid release and Cl- currents in cultured rat astrocytes.J Physiol. 2006 May 1;572(Pt 3):677-89. doi: 10.1113/jphysiol.2005.103820. J Physiol. 2006. PMID: 16527858 Free PMC article.
-
The signaling role for chloride in the bidirectional communication between neurons and astrocytes.Neurosci Lett. 2019 Jan 10;689:33-44. doi: 10.1016/j.neulet.2018.01.012. Epub 2018 Jan 9. Neurosci Lett. 2019. PMID: 29329909 Free PMC article. Review.
-
Volume-regulated anion channel--a frenemy within the brain.Pflugers Arch. 2016 Mar;468(3):421-41. doi: 10.1007/s00424-015-1765-6. Epub 2015 Dec 1. Pflugers Arch. 2016. PMID: 26620797 Free PMC article. Review.
Cited by
-
Astrocytic modulation of potassium under seizures.Neural Regen Res. 2020 Jun;15(6):980-987. doi: 10.4103/1673-5374.270295. Neural Regen Res. 2020. PMID: 31823867 Free PMC article. Review.
-
A Theoretical and Practical Analysis of Membrane Protein Genes Altered in Neutrophils in Parkinson's Disease.Curr Issues Mol Biol. 2025 Jun 13;47(6):459. doi: 10.3390/cimb47060459. Curr Issues Mol Biol. 2025. PMID: 40699858 Free PMC article.
-
Dynamic expression of homeostatic ion channels in differentiated cortical astrocytes in vitro.Pflugers Arch. 2022 Feb;474(2):243-260. doi: 10.1007/s00424-021-02627-x. Epub 2021 Nov 4. Pflugers Arch. 2022. PMID: 34734327 Free PMC article.
-
The Role of Ion-Transporting Proteins on Crosstalk Between the Skeletal Muscle and Central Nervous Systems Elicited by Physical Exercise.Mol Neurobiol. 2025 May;62(5):5546-5565. doi: 10.1007/s12035-024-04613-7. Epub 2024 Nov 22. Mol Neurobiol. 2025. PMID: 39578339 Review.
-
Human iPSC-Derived Astrocytes: A Powerful Tool to Study Primary Astrocyte Dysfunction in the Pathogenesis of Rare Leukodystrophies.Int J Mol Sci. 2021 Dec 27;23(1):274. doi: 10.3390/ijms23010274. Int J Mol Sci. 2021. PMID: 35008700 Free PMC article. Review.

