Prostate Radiotherapy for Metastatic Hormone-sensitive Prostate Cancer: A STOPCAP Systematic Review and Meta-analysis
- PMID: 30826218
- PMCID: PMC6575150
- DOI: 10.1016/j.eururo.2019.02.003
Prostate Radiotherapy for Metastatic Hormone-sensitive Prostate Cancer: A STOPCAP Systematic Review and Meta-analysis
Abstract
Background: Many trials are evaluating therapies for men with metastatic hormone-sensitive prostate cancer (mHSPC).
Objective: To systematically review trials of prostate radiotherapy.
Design, setting, and participants: Using a prospective framework (framework for adaptive meta-analysis [FAME]), we prespecified methods before any trial results were known. We searched extensively for eligible trials and asked investigators when results would be available. We could then anticipate that a definitive meta-analysis of the effects of prostate radiotherapy was possible. We obtained prepublication, unpublished, and harmonised results from investigators.
Intervention: We included trials that randomised men to prostate radiotherapy and androgen deprivation therapy (ADT) or ADT only.
Outcome measurements and statistical analysis: Hazard ratios (HRs) for the effects of prostate radiotherapy on survival, progression-free survival (PFS), failure-free survival (FFS), biochemical progression, and subgroup interactions were combined using fixed-effect meta-analysis.
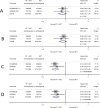
Results and limitations: We identified one ongoing (PEACE-1) and two completed (HORRAD and STAMPEDE) eligible trials. Pooled results of the latter (2126 men; 90% of those eligible) showed no overall improvement in survival (HR=0.92, 95% confidence interval [CI] 0.81-1.04, p=0.195) or PFS (HR=0.94, 95% CI 0.84-1.05, p=0.238) with prostate radiotherapy. There was an overall improvement in biochemical progression (HR=0.74, 95% CI 0.67-0.82, p=0.94×10-8) and FFS (HR=0.76, 95% CI 0.69-0.84, p=0.64×10-7), equivalent to ∼10% benefit at 3yr. The effect of prostate radiotherapy varied by metastatic burden-a pattern consistent across trials and outcome measures, including survival (<5, ≥5; interaction HR=1.47, 95% CI 1.11-1.94, p=0.007). There was 7% improvement in 3-yr survival in men with fewer than five bone metastases.
Conclusions: Prostate radiotherapy should be considered for men with mHSPC with a low metastatic burden.
Patient summary: Prostate cancer that has spread to other parts of the body (metastases) is usually treated with hormone therapy. In men with fewer than five bone metastases, addition of prostate radiotherapy helped them live longer and should be considered.
Keywords: Androgen deprivation therapy; Meta-analysis; Metastases; Prostate cancer; Radiotherapy; Standard of care; Systematic review.
Copyright © 2019 The Authors. Published by Elsevier B.V. All rights reserved.
Figures


Comment in
-
Prostate radiotherapy for metastatic hormone sensitive prostate cancer-myth or reality?Transl Cancer Res. 2019 Nov;8(7):2508-2509. doi: 10.21037/tcr.2019.05.11. Transl Cancer Res. 2019. PMID: 35117005 Free PMC article. No abstract available.
-
Local therapy in patients with metastatic prostate cancer: a new standard of care?Transl Cancer Res. 2019 Dec;8(Suppl 6):S592-S594. doi: 10.21037/tcr.2019.05.27. Transl Cancer Res. 2019. PMID: 35117138 Free PMC article. No abstract available.
References
-
- 2012. STAMPEDE Clinical Trial Protocol V 9.0 (October 2012) http://www.stampedetrial.org/media/1080/mrcctu-stampede-protocol_v90_final_.
-
- Parker C.C., Sydes M.R., Mason M.D. Prostate radiotherapy for men with metastatic disease: a new comparison in the Systemic Therapy in Advancing or Metastatic Prostate Cancer: Evaluation of Drug Efficacy (STAMPEDE) trial. BJU Int. 2013;111:697–699. - PubMed
-
- Fisher D.J. Two-stage individual participant data meta-analysis and generalized forest plots. Stata Journal. 2015;15:369–396.
-
- 2017. StataCorp. Stata Statistical Software: Release 15. College Station, TX, USA: StataCorp LLC.
-
- Tierney J.F., Vale C.L., Burdett S., Fisher D., Rydzewska L.H.M., Parmar M.K.B. Timely and reliable evaluation of the effects of interventions: a framework for adaptive meta-analysis (FAME) Trials. 2017;18(Suppl. 1):P351.
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

