Atrophy in midbrain & cerebral/cerebellar pedunculi is characteristic for progressive supranuclear palsy - A double-validation whole-brain meta-analysis
- PMID: 30831462
- PMCID: PMC6402426
- DOI: 10.1016/j.nicl.2019.101722
Atrophy in midbrain & cerebral/cerebellar pedunculi is characteristic for progressive supranuclear palsy - A double-validation whole-brain meta-analysis
Abstract
Objective: Progressive supranuclear palsy (PSP) is an atypical parkinsonian syndrome characterized by vertical gaze palsy and postural instability. Midbrain atrophy is suggested as a hallmark, but it has not been validated systematically in whole-brain imaging.
Methods: We conducted whole-brain meta-analyses identifying disease-related atrophy in structural MRI. Eighteen studies were identified (N = 315 PSP, 393 controls) and separated into gray or white matter analyses (15/12). All patients were diagnosed according to the National Institute of Neurological Disorders and Stroke and the Society for PSP (NINDS-SPSP criteria, Litvan et al. (1996a)), which are now considered as PSP-Richardson syndrome (Höglinger et al., 2017). With overlay analyses, we double-validated two meta-analytical algorithms: anatomical likelihood estimation and seed-based D mapping. Additionally, we conducted region-of-interest effect size meta-analyses on radiological biomarkers and subtraction analyses differentiating PSP from Parkinson's disease.
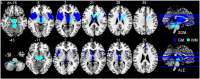
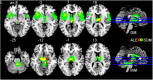
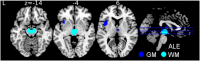
Results: Whole brain meta-analyses revealed consistent gray matter atrophy in bilateral thalamus, anterior insulae, midbrain, and left caudate nucleus. White matter alterations were consistently detected in bilateral superior/middle cerebellar pedunculi, cerebral pedunculi, and midbrain atrophy. Region-of-interest meta-analyses demonstrated that midbrain metrics generally perform very well in distinguishing PSP from other parkinsonian syndromes with strong effect sizes. Subtraction analyses identified the midbrain as differentiating between PSP and Parkinson's disease.
Conclusions: Our meta-analyses identify gray matter atrophy of the midbrain and white matter atrophy of the cerebral/cerebellar pedunculi and midbrain as characteristic for PSP. Results support the incorporation of structural MRI data, and particularly these structures, into the revised PSP diagnostic criteria.
Keywords: Anatomical likelihood estimation; Cerebellar pedunculi; Cerebral pedunculi; Imaging biomarker; Meta-analysis; Midbrain; Progressive supranuclear palsy; Seed-based D mapping.
Copyright © 2019 The Authors. Published by Elsevier Inc. All rights reserved.
Figures

Similar articles
-
Imaging counterpart of postural instability and vertical ocular dysfunction in patients with PSP: A multimodal MRI study.Parkinsonism Relat Disord. 2019 Jun;63:124-130. doi: 10.1016/j.parkreldis.2019.02.022. Epub 2019 Feb 19. Parkinsonism Relat Disord. 2019. PMID: 30803901
-
Beyond the midbrain atrophy: wide spectrum of structural MRI finding in cases of pathologically proven progressive supranuclear palsy.Neuroradiology. 2017 May;59(5):431-443. doi: 10.1007/s00234-017-1812-4. Epub 2017 Apr 6. Neuroradiology. 2017. PMID: 28386688 Review.
-
Midbrain atrophy in patients with presymptomatic progressive supranuclear palsy-Richardson's syndrome.Parkinsonism Relat Disord. 2019 Sep;66:80-86. doi: 10.1016/j.parkreldis.2019.07.009. Epub 2019 Jul 8. Parkinsonism Relat Disord. 2019. PMID: 31307918
-
Patterns of brain atrophy that differentiate corticobasal degeneration syndrome from progressive supranuclear palsy.Arch Neurol. 2006 Jan;63(1):81-6. doi: 10.1001/archneur.63.1.81. Arch Neurol. 2006. PMID: 16401739
-
Gray matter atrophy in progressive supranuclear palsy: meta-analysis of voxel-based morphometry studies.Neurol Sci. 2013 Jul;34(7):1049-55. doi: 10.1007/s10072-013-1406-9. Epub 2013 Mar 30. Neurol Sci. 2013. PMID: 23543378 Review.
Cited by
-
The neuroscience of social feelings: mechanisms of adaptive social functioning.Neurosci Biobehav Rev. 2021 Sep;128:592-620. doi: 10.1016/j.neubiorev.2021.05.028. Epub 2021 Jun 2. Neurosci Biobehav Rev. 2021. PMID: 34089764 Free PMC article. Review.
-
Structural parameters are superior to eigenvector centrality in detecting progressive supranuclear palsy with machine learning & multimodal MRI.Heliyon. 2024 Jul 25;10(15):e34910. doi: 10.1016/j.heliyon.2024.e34910. eCollection 2024 Aug 15. Heliyon. 2024. PMID: 39170550 Free PMC article.
-
Clinical Spectrum of Tauopathies.Front Neurol. 2022 Jul 14;13:944806. doi: 10.3389/fneur.2022.944806. eCollection 2022. Front Neurol. 2022. PMID: 35911892 Free PMC article. Review.
-
Cognitive impairment in Parkinson's disease and other parkinsonian syndromes.J Neural Transm (Vienna). 2025 Mar;132(3):341-355. doi: 10.1007/s00702-024-02865-0. Epub 2024 Nov 30. J Neural Transm (Vienna). 2025. PMID: 39614911 Review.
-
Histologic lesion type correlates of magnetic resonance imaging biomarkers in four-repeat tauopathies.Brain Commun. 2022 Apr 28;4(3):fcac108. doi: 10.1093/braincomms/fcac108. eCollection 2022. Brain Commun. 2022. PMID: 35663380 Free PMC article.
References
-
- Bisenius S., Neumann J., Schroeter M.L. Validating new diagnostic imaging criteria for primary progressive aphasia via anatomical likelihood estimation meta-analyses. Eur. J. Neurol. 2016;23:704–712. - PubMed
-
- Bisenius S., Mueller K., Diehl-Schmid J., Fassbender K., Grimmer T., Jessen F., Kassubek J., Kornhuber J., Landwehrmeyer B., Ludolph A., Schneider A., Anderl-Straub S., Stuke K., Danek A., Otto M., Schroeter M.L., group F.T.s. Predicting primary progressive aphasias with support vector machine approaches in structural MRI data. Neuroimage Clin. 2017;14:334–343. - PMC - PubMed
-
- Boxer A.L., Geschwind M.D., Belfor N. Patterns of brain atrophy that differentiate corticobasal degeneration syndrome from progressive supranuclear palsy. Arch. Neurol. 2006;63(1):81–86. - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

