Evaluation of the Liver Toxicity of Pterocephalus hookeri Extract via Triggering Necrosis
- PMID: 30832306
- PMCID: PMC6468695
- DOI: 10.3390/toxins11030142
Evaluation of the Liver Toxicity of Pterocephalus hookeri Extract via Triggering Necrosis
Abstract
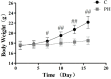
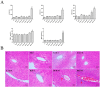
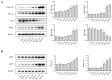
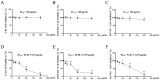
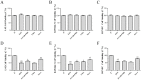
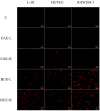
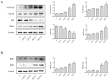
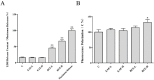
Pterocephalus hookeri (C. B. Clarke) Höeck, recorded in the Chinese Pharmacopoeia (2015 version) as a Tibetan medicine for the treatment of various diseases, especially rheumatoid arthritis, was believed to possess a slight toxicity. However, hardly any research has been carried out about it. The present study aimed to evaluate the toxicity in vivo and in vitro. Toxicity was observed by the evaluation of mice weight loss and histopathological changes in the liver. Then, the comparison research between ethyl acetate extract (EAE) and n-butanol extract (BUE) suggested that liver toxicity was mainly induced by BUE. The mechanical study suggested that BUE-induced liver toxicity was closely associated with necrosis detected by MTT and propidium iodide (PI) staining, via releasing lactate dehydrogenase (LDH), reducing the fluidity, and increasing the permeability of the cell membrane. Western blot analysis confirmed that the necrosis occurred molecularly by the up-regulation of receptor-interacting protein kinase 1 (RIP1) and receptor-interacting protein kinase 3 (RIP3), as well as the activation of the nuclear factor-kappa-gene binding (NF-κB) signaling pathway in vivo and in vitro. This finding indicated that the liver toxicity induced by BUE from P. hookeri was mainly caused by necrosis, which provides an important theoretical support for further evaluation of the safety of this folk medicine.
Keywords: Pterocephalus hookeri; inflammation; liver toxicity; n-butanol extract; necrosis.
Conflict of interest statement
The authors declare no conflict of interest.
Figures

Similar articles
-
Traditional uses, phytochemistry, pharmacology, and toxicology of Pterocephalus hookeri (C. B. Clarke) Höeck: a review.RSC Adv. 2021 Aug 26;11(46):28761-28774. doi: 10.1039/d1ra05548h. eCollection 2021 Aug 23. RSC Adv. 2021. PMID: 35478563 Free PMC article. Review.
-
Pterocephin A, a novel Triterpenoid Saponin from Pterocephalus hookeri induced liver injury by activation of necroptosis.Phytomedicine. 2021 May;85:153548. doi: 10.1016/j.phymed.2021.153548. Epub 2021 Mar 19. Phytomedicine. 2021. PMID: 33831690
-
The anti-arthritic activity of total glycosides from Pterocephalus hookeri, a traditional Tibetan herbal medicine.Pharm Biol. 2017 Dec;55(1):560-570. doi: 10.1080/13880209.2016.1263869. Pharm Biol. 2017. PMID: 27937009 Free PMC article.
-
Palmitate induces RIP1/RIP3-dependent necrosis via MLKL-mediated pore formation in the plasma membrane of RAW 264.7 cells.Biochem Biophys Res Commun. 2017 Jan 8;482(2):359-365. doi: 10.1016/j.bbrc.2016.11.068. Epub 2016 Nov 14. Biochem Biophys Res Commun. 2017. PMID: 27856241
-
Final report on the safety assessment of capsicum annuum extract, capsicum annuum fruit extract, capsicum annuum resin, capsicum annuum fruit powder, capsicum frutescens fruit, capsicum frutescens fruit extract, capsicum frutescens resin, and capsaicin.Int J Toxicol. 2007;26 Suppl 1:3-106. doi: 10.1080/10915810601163939. Int J Toxicol. 2007. PMID: 17365137 Review.
Cited by
-
Oxybaphus himalaicus Mitigates Lipopolysaccharide-Induced Acute Kidney Injury by Inhibiting TLR4/MD2 Complex Formation.Antioxidants (Basel). 2022 Nov 22;11(12):2307. doi: 10.3390/antiox11122307. Antioxidants (Basel). 2022. PMID: 36552516 Free PMC article.
-
Toxicokinetics and Tissue Distribution of the Hepatotoxic Triterpenoid Saponin Pterocephin A in Rats Using the Ultra-Performance Liquid Chromatography-Tandem Mass Spectrometry (UPLC-MS/MS) Method.Molecules. 2024 Oct 25;29(21):5044. doi: 10.3390/molecules29215044. Molecules. 2024. PMID: 39519684 Free PMC article.
-
Traditional uses, phytochemistry, pharmacology, and toxicology of Pterocephalus hookeri (C. B. Clarke) Höeck: a review.RSC Adv. 2021 Aug 26;11(46):28761-28774. doi: 10.1039/d1ra05548h. eCollection 2021 Aug 23. RSC Adv. 2021. PMID: 35478563 Free PMC article. Review.
References
-
- Xia Q., Zhang X., Xu K., Zhang W., Fan J., Zhao C., Feng Y., Ni Y., Ma Z., Lin R. Review on toxicity of toxic traditional Chinese medicine recorded in Chinese pharmacopoeia (2015 version) Glob. Tradit. Chin. Med. 2017:377–384. doi: 10.3969/j.issn.1674-1749.2017.03.037. - DOI
-
- Ng A.W.T., Poon S.L., Huang M.N., Lim J.Q., Boot A., Yu W., Suzuki Y., Thangaraju S., Ng C.C.Y., Tan P., et al. Aristolochic acids and their derivatives are widely implicated in liver cancers in Taiwan and throughout Asia. Sci. Transl. Med. 2017;9:eaan6446. doi: 10.1126/scitranslmed.aan6446. - DOI - PubMed
-
- Wang J., Ma Z., Niu M., Zhu Y., Liang Q., Zhao Y., Song J., Bai Z., Zhang Y., Zhang P., et al. Evidence chain-based causality identification in herb-induced liver injury: Exemplification of a well-known liver-restorative herb Polygonum multiflorum. Front. Med. 2015;9:457–467. doi: 10.1007/s11684-015-0417-8. - DOI - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Research Materials
Miscellaneous

