Control of Staphylococcus aureus Quorum Sensing by a Membrane-Embedded Peptidase
- PMID: 30833334
- PMCID: PMC6479040
- DOI: 10.1128/IAI.00019-19
Control of Staphylococcus aureus Quorum Sensing by a Membrane-Embedded Peptidase
Abstract
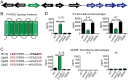
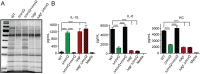
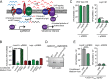
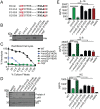
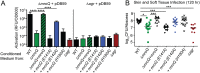
Gram-positive bacteria process and release small peptides, or pheromones, that act as signals for the induction of adaptive traits, including those involved in pathogenesis. One class of small signaling pheromones is the cyclic autoinducing peptides (AIPs), which regulate expression of genes that orchestrate virulence and persistence in a range of microbes, including staphylococci, listeriae, clostridia, and enterococci. In a genetic screen for Staphylococcus aureus secreted virulence factors, we identified an S. aureus mutant containing an insertion in the gene SAUSA300_1984 (mroQ), which encodes a putative membrane-embedded metalloprotease. A ΔmroQ mutant exhibited impaired induction of Toll-like receptor 2-dependent inflammatory responses from macrophages but elicited greater production of the inflammatory cytokine interleukin-1β and was attenuated in a murine skin and soft tissue infection model. The ΔmroQ mutant phenocopies an S. aureus mutant containing a deletion of the accessory gene regulatory system (Agr), wherein both strains have significantly reduced production of secreted toxins and virulence factors but increased surface protein A abundance. The Agr system controls virulence factor gene expression in S. aureus by sensing the accumulation of AIP via the histidine kinase AgrC and the response regulator AgrA. We provide evidence to suggest that MroQ acts within the Agr pathway to facilitate the optimal processing or export of AIP for signal amplification through AgrC/A and induction of virulence factor gene expression. Mutation of MroQ active-site residues significantly reduces AIP signaling and attenuates virulence. Altogether, this work identifies a new component of the Agr quorum-sensing circuit that is critical for the production of S. aureus virulence factors.
Keywords: Agr; Staphylococcus aureus; accessory gene regulator; peptidase; peptide signaling; quorum sensing; virulence.
Copyright © 2019 American Society for Microbiology.
Figures


Similar articles
-
Characterization of MroQ-Dependent Maturation and Export of the Staphylococcus aureus Accessory Gene Regulatory System Autoinducing Peptide.Infect Immun. 2022 Oct 20;90(10):e0026322. doi: 10.1128/iai.00263-22. Epub 2022 Sep 8. Infect Immun. 2022. PMID: 36073934 Free PMC article.
-
Reconstitution of the S. aureus agr quorum sensing pathway reveals a direct role for the integral membrane protease MroQ in pheromone biosynthesis.Proc Natl Acad Sci U S A. 2022 Aug 16;119(33):e2202661119. doi: 10.1073/pnas.2202661119. Epub 2022 Aug 8. Proc Natl Acad Sci U S A. 2022. PMID: 35939668 Free PMC article.
-
Timing Is Everything: Impact of Naturally Occurring Staphylococcus aureus AgrC Cytoplasmic Domain Adaptive Mutations on Autoinduction.J Bacteriol. 2019 Sep 20;201(20):e00409-19. doi: 10.1128/JB.00409-19. Print 2019 Oct 15. J Bacteriol. 2019. PMID: 31358609 Free PMC article.
-
Quorum-sensing, intra- and inter-species competition in the staphylococci.Microbiology (Reading). 2023 Aug;169(8):001381. doi: 10.1099/mic.0.001381. Microbiology (Reading). 2023. PMID: 37578829 Free PMC article. Review.
-
Quorum sensing-mediated regulation of staphylococcal virulence and antibiotic resistance.Future Microbiol. 2014;9(5):669-81. doi: 10.2217/fmb.14.31. Future Microbiol. 2014. PMID: 24957093 Review.
Cited by
-
Staphylococcus aureus adapts to the host nutritional landscape to overcome tissue-specific branched-chain fatty acid requirement.Proc Natl Acad Sci U S A. 2021 Mar 30;118(13):e2022720118. doi: 10.1073/pnas.2022720118. Proc Natl Acad Sci U S A. 2021. PMID: 33753501 Free PMC article.
-
Evolution of two-component quorum sensing systems.Access Microbiol. 2022 Jan 12;4(1):000303. doi: 10.1099/acmi.0.000303. eCollection 2022. Access Microbiol. 2022. PMID: 35252749 Free PMC article.
-
Characterization of MroQ-Dependent Maturation and Export of the Staphylococcus aureus Accessory Gene Regulatory System Autoinducing Peptide.Infect Immun. 2022 Oct 20;90(10):e0026322. doi: 10.1128/iai.00263-22. Epub 2022 Sep 8. Infect Immun. 2022. PMID: 36073934 Free PMC article.
-
Chemical and biomolecular insights into the Staphylococcus aureus agr quorum sensing system: Current progress and ongoing challenges.Isr J Chem. 2023 Jun;63(5-6):e202200096. doi: 10.1002/ijch.202200096. Epub 2023 Mar 16. Isr J Chem. 2023. PMID: 38765792 Free PMC article.
-
Comparison of Antivirulence Activities of Black Ginseng against Methicillin-Resistant Staphylococcus aureus According to the Number of Repeated Steaming and Drying Cycles.Antibiotics (Basel). 2021 May 21;10(6):617. doi: 10.3390/antibiotics10060617. Antibiotics (Basel). 2021. PMID: 34064076 Free PMC article.
References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Molecular Biology Databases
Miscellaneous

