Risk Factors for Retinopathy in Type 1 Diabetes: The DCCT/EDIC Study
- PMID: 30833368
- PMCID: PMC6489114
- DOI: 10.2337/dc18-2308
Risk Factors for Retinopathy in Type 1 Diabetes: The DCCT/EDIC Study
Abstract
Objective: The Diabetes Control and Complications Trial (DCCT) demonstrated that intensive therapy reduced the development and progression of retinopathy in type 1 diabetes (T1D) compared with conventional therapy. The Epidemiology of Diabetes Interventions and Complications (EDIC) study observational follow-up showed persistent benefits. In addition to glycemia, we now examine other potential retinopathy risk factors (modifiable and nonmodifiable) over more than 30 years of follow-up in DCCT/EDIC.
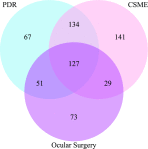
Research design and methods: The retinopathy outcomes were proliferative diabetic retinopathy (PDR), clinically significant macular edema (CSME), and ocular surgery. The survival (event-free) probability was estimated using the Kaplan-Meier method. Cox proportional hazards models assessed the association between risk factors and subsequent risk of retinopathy. Both forward- and backward-selection approaches determined the multivariable models.
Results: Rate of ocular events per 1,000 person-years was 12 for PDR, 14.5 for CSME, and 7.6 for ocular surgeries. Approximately 65%, 60%, and 70% of participants remained free of PDR, CSME, and ocular surgery, respectively. The greatest risk factors for PDR in descending order were higher mean HbA1c, longer duration of T1D, elevated albumin excretion rate (AER), and higher mean diastolic blood pressure (DBP). For CSME, risk factors, in descending order, were higher mean HbA1c, longer duration of T1D, and greater age and DBP and, for ocular surgeries, were higher mean HbA1c, older age, and longer duration of T1D.
Conclusions: Mean HbA1c was the strongest risk factor for the progression of retinopathy. Although glycemic control is important, elevated AER and DBP were other modifiable risk factors associated with the progression of retinopathy.
Trial registration: ClinicalTrials.gov NCT00360893 NCT00360815.
© 2019 by the American Diabetes Association.
Figures
References
-
- Nathan DM, Genuth S, Lachin J, et al.; Diabetes Control and Complications Trial Research Group . The effect of intensive treatment of diabetes on the development and progression of long-term complications in insulin-dependent diabetes mellitus. N Engl J Med 1993;329:977–986 - PubMed
-
- Lachin JM, Genuth S, Cleary P, Davis MD, Nathan DM; Diabetes Control and Complications Trial/Epidemiology of Diabetes Interventions and Complications Research Group . Retinopathy and nephropathy in patients with type 1 diabetes four years after a trial of intensive therapy. N Engl J Med 2000;342:381–389 - PMC - PubMed
-
- Lachin JM, White NH, Hainsworth DP, et al.; Diabetes Control and Complications Trial (DCCT)/Epidemiology of Diabetes Interventions and Complications (EDIC) Research Group . Effect of intensive diabetes therapy on the progression of diabetic retinopathy in patients with type 1 diabetes: 18 years of follow-up in the DCCT/EDIC. Diabetes 2015;64:631–642 - PMC - PubMed
Publication types
MeSH terms
Substances
Associated data
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous