Effect of methylprednisolone on acute kidney injury in patients undergoing cardiac surgery with a cardiopulmonary bypass pump: a randomized controlled trial
- PMID: 30833491
- PMCID: PMC6400656
- DOI: 10.1503/cmaj.181644
Effect of methylprednisolone on acute kidney injury in patients undergoing cardiac surgery with a cardiopulmonary bypass pump: a randomized controlled trial
Abstract
Background: Perioperative corticosteroid use may reduce acute kidney injury. We sought to test whether methylprednisolone reduces the risk of acute kidney injury after cardiac surgery.
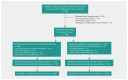
Methods: We conducted a prespecified substudy of a randomized controlled trial involving patients undergoing cardiac surgery with cardiopulmonary bypass (2007-2014); patients were recruited from 79 centres in 18 countries. Eligibility criteria included a moderate-to-high risk of perioperative death based on a preoperative score of 6 or greater on the European System for Cardiac Operative Risk Evaluation I. Patients (n = 7286) were randomly assigned (1:1) to receive intravenous methylprednisolone (250 mg at anesthetic induction and 250 mg at initiation of cardiopulmonary bypass) or placebo. Patients, caregivers, data collectors and outcome adjudicators were unaware of the assigned intervention. The primary outcome was postoperative acute kidney injury, defined as an increase in the serum creatinine concentration (from the preoperative value) of 0.3 mg/dL or greater (≥ 26.5 μmol/L) or 50% or greater in the 14-day period after surgery, or use of dialysis within 30 days after surgery.
Results: Acute kidney injury occurred in 1479/3647 patients (40.6%) in the methylprednisolone group and in 1426/3639 patients (39.2%) in the placebo group (adjusted relative risk 1.04, 95% confidence interval 0.96 to 1.11). Results were consistent across several definitions of acute kidney injury and in patients with preoperative chronic kidney disease.
Interpretation: Intraoperative corticosteroid use did not reduce the risk of acute kidney injury in patients with a moderate-to-high risk of perioperative death who had cardiac surgery with cardiopulmonary bypass. Our results do not support the prophylactic use of steroids during cardiopulmonary bypass surgery. Trial registration: ClinicalTrials.gov, no. NCT00427388.
© 2019 Joule Inc. or its licensors.
Conflict of interest statement
Competing interests: P.J. Devereaux has received grants from Abbott Diagnostics, Boehringer Ingelheim, Covidien, Octapharma, Philips Healthcare, Roche Diagnostics and Stryker for projects outside the work reported here. Chirag Parikh reports personal fees from AbbVie Pharmaceutical Research and Development and GENFIT Biopharmaceutical Company; funding from Renalytix AI; and grants from the National Institute of Diabetes and Digestive and Kidney Diseases, and the National Heart, Lung and Blood Institute. Vlado Perkovic reports receiving personal fees for advisory boards or scientific presentations from Retrophin, Janssen, Merck and Servier. Vlado Perkovic was a member of the SONAR Steering Committee; has served on steering committees for trials funded by AbbVie, Boehringer Ingelheim, GlaxoSmithKline, Janssen, Novo Nordisk, Retrophin and Tricida; and has participated in scientific presentations and advisory boards with AbbVie, Astellas, Astra-Zeneca, Bayer, Baxter, Bristol-Myers Squibb, Boehringer Ingelheim, Dimerix, Durect, Eli Lilly, Gilead, GlaxoSmithKline, Janssen, Merck, Mitsubishi Tanabe, Novartis, Novo Nordisk, Pfizer, PharmaLink, Relypsa, Retrophin, Sanofi, Servier, Vifor and Tricida. Richard Whitlock reports grants and personal fees from Boehringer Ingelheim. No other competing interests were declared.
Figures

References
-
- Rosner MH, Okusa MD. Acute kidney injury associated with cardiac surgery. Clin J Am Soc Nephrol 2006;1:19–32. - PubMed
-
- Chertow GM, Burdick E, Honour M, et al. Acute kidney injury, mortality, length of stay, and costs in hospitalized patients. J Am Soc Nephrol 2005;16:3365–70. - PubMed
-
- Dieleman JM, Nierich AP, Rosseel PM, et al. Dexamethasone for Cardiac Surgery (DECS) Study Group. Intraoperative high-dose dexamethasone for cardiac surgery: a randomized controlled trial. JAMA 2012;308:1761–7. - PubMed
Publication types
MeSH terms
Substances
Associated data
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
