Animal Models of (or for) Aggression Reward, Addiction, and Relapse: Behavior and Circuits
- PMID: 30833504
- PMCID: PMC6529864
- DOI: 10.1523/JNEUROSCI.0151-19.2019
Animal Models of (or for) Aggression Reward, Addiction, and Relapse: Behavior and Circuits
Abstract
Inappropriate and pathological aggression plays a leading role in the suffering and death of millions of people, and further places an untenable strain on the caregivers and families of those afflicted. In some cases, such as addictive drugs, aggression can be highly rewarding (appetitive) and continually pursued despite short- and long-term negative consequences. Similarly, recidivism (relapse) rates for repeat violent offenders are as high as relapse rates for drug addicts. Appetitive aggression and relapse to aggression seeking can be modeled in mice studies using conditioned place preference and self-administration procedures followed by a period of abstinence and subsequent tests for relapse to aggression preference and aggression seeking. These procedures allow for the study of the mechanisms that control the appetitive versus the consummatory (attack) phases of aggressive behavior. In this review, we first discuss the behavioral procedures developed to probe appetitive aggression in mouse models, spanning from Pavlovian to operant tasks, and we also describe the recently proposed phenomenon of "aggression addiction." Next, we discuss the pharmacological and circuit mechanisms of aggression conditioned place preference and aggression self-administration, seeking, and relapse, highlighting mechanistic congruence and divergence between appetitive and consummatory phases of aggression. We conclude by discussing clinical implications of the studies reviewed.
Copyright © 2019 the authors.
Figures





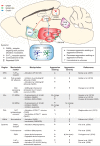
References
-
- Aleyasin H, Flanigan ME, Golden SA, Takahashi A, Menard C, Pfau ML, Multer J, Pina J, McCabe KA, Bhatti N, Hodes GE, Heshmati M, Neve RL, Nestler EJ, Heller EA, Russo SJ (2018b) Cell-type-specific role of DeltaFosB in nucleus accumbens in modulating intermale aggression. J Neurosci 38:5913–5924. 10.1523/JNEUROSCI.0296-18.2018 - DOI - PMC - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
