The Skeletal Muscle as an Active Player Against Cancer Cachexia
- PMID: 30833900
- PMCID: PMC6387914
- DOI: 10.3389/fphys.2019.00041
The Skeletal Muscle as an Active Player Against Cancer Cachexia
Abstract
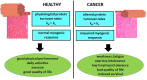
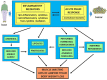
The management of cancer patients is frequently complicated by the occurrence of cachexia. This is a complex syndrome that markedly impacts on quality of life as well as on tolerance and response to anticancer treatments. Loss of body weight, wasting of both adipose tissue and skeletal muscle and reduced survival rates are among the main features of cachexia. Skeletal muscle wasting has been shown to depend, mainly at least, on the induction of protein degradation rates above physiological levels. Such hypercatabolic pattern is driven by overactivation of different intracellular proteolytic systems, among which those dependent on ubiquitin-proteasome and autophagy. Selective rather than bulk degradation of altered proteins and organelles was also proposed to occur. Within the picture described above, the muscle is frequently considered a sort of by-stander tissue where external stimuli, directly or indirectly, can poise protein metabolism toward a catabolic setting. By contrast, several observations suggest that the muscle reacts to the wasting drive imposed by cancer growth by activating different compensatory strategies that include anabolic capacity, the activation of autophagy and myogenesis. Even if muscle response is eventually ill-fated, its occurrence supports the idea that in the presence of appropriate treatments the development of cancer-induced wasting might not be an ineluctable event in tumor hosts.
Keywords: adaptive response; energy metabolism; muscle wasting; myogenesis; oxidative stress; protein turnover.
Figures

References
Publication types
LinkOut - more resources
Full Text Sources
Other Literature Sources

