Modulation of Renal Parenchyma in Response to Allogeneic Adipose-Derived Mesenchymal Stem Cells Transplantation in Acute Kidney Injury
- PMID: 30836723
- PMCID: PMC6457705
- DOI: 10.15283/ijsc18091
Modulation of Renal Parenchyma in Response to Allogeneic Adipose-Derived Mesenchymal Stem Cells Transplantation in Acute Kidney Injury
Abstract
Background and objectives: In regenerative medicine, mesenchymal stem cells derived from adipose tissues (Ad-MSCs) are a very attractive target to treat many diseases. In relation to nephrology, the aim of the current study is to investigate the effects of Ad-MSCs for the amelioration of acute kidney injury and to explore the mechanism of renal parenchymal changes in response to allogeneic transplantation of Ad-MSCs.
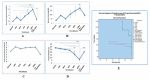
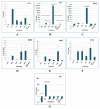
Methods and results: The nephrotoxicity was induced by cisplatin (CP) in balb/c mice according to RIFLE Class and AKIN Stage 3. PCR, qRT-PCR and fluorescent labeled cells infusion, histopathology, immunohistochemistry, functional analyses were used for genes and proteins expressions data acquisition respectively. We demonstrated that single intravenous infusion of 2.5×107/kg mAd-MSCs in mice pre-injected with CP recruited to the kidney, restored the renal structure, and function, which resulted in progressive survival of mice. The renal tissue morphology was recovered in terms of diminished necrosis or epithelial cells damage, protein casts formation, infiltration of inflammatory cells, tubular dilatation, and restoration of brush border protein; Megalin and decreased Kim-1 expressions in mAd-MSCs transplanted mice. Significant reduction in serum creatinine with slashed urea and urinary protein levels were observed. Anti-BrdU staining displayed enhanced tubular cells proliferation. Predominantly, downgrade expressions of TNF-α and TGF-β1 were observed post seven days in mAd-MSCs transplanted mice.
Conclusions: Ad-MSCs exerts pro-proliferative, anti-inflammatory, and anti-fibrotic effects. Ad-MSCs transplantation without any chemical or genetic manipulation can provide the evidence of therapeutic strategy for the origin of regeneration and overall an improved survival of the system in functionally deprived failed kidneys.
Keywords: Acute kidney injury; Adipose-derived mesenchymal stem cells; Cisplatin; TGF-β1; TNF-α; Tubular injury.
Conflict of interest statement
The authors have no conflicting financial interest.
Figures



Similar articles
-
Human adipose tissue-derived mesenchymal stem cells protect kidneys from cisplatin nephrotoxicity in rats.Am J Physiol Renal Physiol. 2012 May 1;302(9):F1141-50. doi: 10.1152/ajprenal.00060.2011. Epub 2011 Dec 28. Am J Physiol Renal Physiol. 2012. PMID: 22205231
-
Comparative study of allogenic and xenogeneic mesenchymal stem cells on cisplatin-induced acute kidney injury in Sprague-Dawley rats.Stem Cell Res Ther. 2016 Sep 1;7(1):126. doi: 10.1186/s13287-016-0386-0. Stem Cell Res Ther. 2016. PMID: 27585525 Free PMC article.
-
Autologous bone marrow-derived mesenchymal stem cells for interstitial fibrosis and tubular atrophy: a pilot study.Ren Fail. 2021 Dec;43(1):1266-1275. doi: 10.1080/0886022X.2021.1968432. Ren Fail. 2021. PMID: 34493167 Free PMC article.
-
Human adipose-derived mesenchymal stem cells repair cisplatin-induced acute kidney injury through antiapoptotic pathways.Exp Ther Med. 2015 Aug;10(2):468-476. doi: 10.3892/etm.2015.2505. Epub 2015 May 21. Exp Ther Med. 2015. PMID: 26622339 Free PMC article.
-
Regenerative pharmacology for the treatment of acute kidney injury: Skeletal muscle stem/progenitor cells for renal regeneration?Pharmacol Res. 2016 Nov;113(Pt B):802-807. doi: 10.1016/j.phrs.2016.03.014. Epub 2016 Mar 18. Pharmacol Res. 2016. PMID: 27001227 Review.
Cited by
-
Culturing Conditions Dictate the Composition and Pathways Enrichment of Human and Rat Perirenal Adipose-Derived Stromal Cells' Secretomes.Stem Cell Rev Rep. 2024 Oct;20(7):1869-1888. doi: 10.1007/s12015-024-10748-w. Epub 2024 Jun 26. Stem Cell Rev Rep. 2024. PMID: 38922529 Free PMC article.
-
Mesenchymal Stem Cell Therapy in Kidney Diseases: Potential and Challenges.Cell Transplant. 2023 Jan-Dec;32:9636897231164251. doi: 10.1177/09636897231164251. Cell Transplant. 2023. PMID: 37013255 Free PMC article. Review.
-
Mesenchymal stromal cells: what have we learned so far about their therapeutic potential and mechanisms of action?Emerg Top Life Sci. 2021 Oct 29;5(4):549-562. doi: 10.1042/ETLS20210013. Emerg Top Life Sci. 2021. PMID: 34495324 Free PMC article. Review.
References
-
- Morigi M, Imberti B, Zoja C, Corna D, Tomasoni S, Abbate M, Rottoli D, Angioletti S, Benigni A, Perico N, Alison M, Remuzzi G. Mesenchymal stem cells are renotropic, helping to repair the kidney and improve function in acute renal failure. J Am Soc Nephrol. 2004;15:1794–1804. doi: 10.1097/01.ASN.0000128974.07460.34. - DOI - PubMed
LinkOut - more resources
Full Text Sources
Miscellaneous

