Ceria-Based Materials in Hydrogenation and Reforming Reactions for CO2 Valorization
- PMID: 30838198
- PMCID: PMC6382745
- DOI: 10.3389/fchem.2019.00028
Ceria-Based Materials in Hydrogenation and Reforming Reactions for CO2 Valorization
Abstract
Reducing greenhouse emissions is of vital importance to tackle the climate changes and to decrease the carbon footprint of modern societies. Today there are several technologies that can be applied for this goal and especially there is a growing interest in all the processes dedicated to manage CO2 emissions. CO2 can be captured, stored or reused as carbon source to produce chemicals and fuels through catalytic technologies. This study reviews the use of ceria based catalysts in some important CO2 valorization processes such as the methanation reaction and methane dry-reforming. We analyzed the state of the art with the aim of highlighting the distinctive role of ceria in these reactions. The presence of cerium based oxides generally allows to obtain a strong metal-support interaction with beneficial effects on the dispersion of active metal phases, on the selectivity and durability of the catalysts. Moreover, it introduces different functionalities such as redox and acid-base centers offering versatility of approaches in designing and engineering more powerful formulations for the catalytic valorization of CO2 to fuels.
Keywords: CO2 methanation; CO2 valorization; CeO2; ceria based oxides; gas to fuel technologies; methane dry reforming to syngas.
Figures

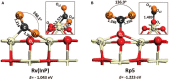

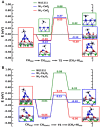
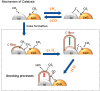
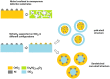

References
-
- Abasaeed A. E., Al-Fatesh A. S., Naeem M. A., Ibrahim A. A., Fakeeha A. H. (2015). Catalytic performance of CeO2 and ZrO2 supported Co catalysts for hydrogen production via dry reforming of methane. Int. J. Hydrog. Energy 40, 6818–6826. 10.1016/j.ijhydene.2015.03.152s - DOI
-
- Abate S., Mebrahtu C., Giglio E., Deorsola F., Bensaid S., Perathoner S., et al. (2016). Catalytic performance of γ-Al2O3-ZrO2-TiO2-CeO2 composite oxide supported Ni-based catalysts for CO2 methanation. Ind. Eng. Chem. Res. 55, 4451–4460. 10.1021/acs.iecr.6b00134 - DOI
-
- Aldana P. A. U., Ocampo F., Kobl K., Louis B., Thibault-Starzyk F., Daturi M., et al. (2013). Catalytic CO2 valorization into CH4 on Ni-based ceria-zirconia. reaction mechanism by operando IR spectroscopy. Catal. Today 215, 201–207. 10.1016/j.cattod.2013.02.019 - DOI
-
- Alvarez A., Bansode A., Urakawa A., Bavykina A. V., Wezendonk T. A., Makkee M., et al. . (2017). Challenges in the greener production of formates/formic acid, methanol, and DME by heterogeneously catalyzed CO2 hydrogenation processes. Chem. Rev. 117, 9804–9838. 10.1021/acs.chemrev.6b00816 - DOI - PMC - PubMed
Publication types
LinkOut - more resources
Full Text Sources

