Macroencapsulated Human iPSC-Derived Pancreatic Progenitors Protect against STZ-Induced Hyperglycemia in Mice
- PMID: 30853374
- PMCID: PMC6449839
- DOI: 10.1016/j.stemcr.2019.02.002
Macroencapsulated Human iPSC-Derived Pancreatic Progenitors Protect against STZ-Induced Hyperglycemia in Mice
Abstract
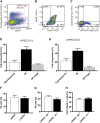
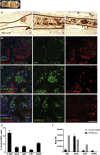
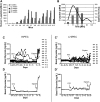
In type 1 diabetes, a renewable source of human pancreatic β cells, in particular from human induced pluripotent stem cell (hiPSC) origin, would greatly benefit cell therapy. Earlier work showed that pancreatic progenitors differentiated from human embryonic stem cells in vitro can further mature to become glucose responsive following macroencapsulation and transplantation in mice. Here we took a similar approach optimizing the generation of pancreatic progenitors from hiPSCs. This work demonstrates that hiPSCs differentiated to pancreatic endoderm in vitro can be efficiently and robustly generated under large-scale conditions. The hiPSC-derived pancreatic endoderm cells (HiPECs) can further differentiate into glucose-responsive islet-like cells following macroencapsulation and in vivo implantation. The HiPECs can protect mice from streptozotocin-induced hyperglycemia and maintain normal glucose homeostasis and equilibrated plasma glucose concentrations at levels similar to the human set point. These results further validate the potential use of hiPSC-derived islet cells for application in clinical settings.
Keywords: diabetes mellitus; differentiation; encapsulation; human; iPSC; stem cell; therapy; β cell.
Copyright © 2019 The Authors. Published by Elsevier Inc. All rights reserved.
Figures




References
-
- Castaing M., Peault B., Basmaciogullari A., Casal I., Czernichow P., Scharfmann R. Blood glucose normalization upon transplantation of human embryonic pancreas into beta-cell-deficient SCID mice. Diabetologia. 2001;44:2066–2076. - PubMed
-
- D'Amour K.A., Agulnick A.D., Eliazer S., Kelly O.G., Kroon E., Baetge E.E. Efficient differentiation of human embryonic stem cells to definitive endoderm. Nat. Biotechnol. 2005;23:1534–1541. - PubMed
-
- D'Amour K.A., Bang A.G., Eliazer S., Kelly O.G., Agulnick A.D., Smart N.G., Moorman M.A., Kroon E., Carpenter M.K., Baetge E.E. Production of pancreatic hormone-expressing endocrine cells from human embryonic stem cells. Nat. Biotechnol. 2006;24:1392–1401. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

