Biosynthesized Silver Nanoparticle (AgNP) From Pandanus odorifer Leaf Extract Exhibits Anti-metastasis and Anti-biofilm Potentials
- PMID: 30853939
- PMCID: PMC6396724
- DOI: 10.3389/fmicb.2019.00008
Biosynthesized Silver Nanoparticle (AgNP) From Pandanus odorifer Leaf Extract Exhibits Anti-metastasis and Anti-biofilm Potentials
Abstract
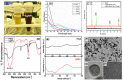
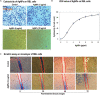
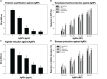
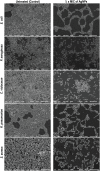
Cancer and the associated secondary bacterial infections are leading cause of mortality, due to the paucity of effective drugs. Here, we have synthesized silver nanoparticles (AgNPs) from organic resource and confirmed their anti-cancer and anti-microbial potentials. Microwave irradiation method was employed to synthesize AgNPs using Pandanus odorifer leaf extract. Anti-cancer potential of AgNPs was evaluated by scratch assay on the monolayer of rat basophilic leukemia (RBL) cells, indicating that the synthesized AgNPs inhibit the migration of RBL cells. The synthesized AgNPs showed MIC value of 4-16 μg/mL against both Gram +ve and Gram -ve bacterial strains, exhibiting the anti-microbial potential. Biofilm inhibition was recorded at sub-MIC values against Gram +ve and Gram -ve bacterial strains. Violacein and alginate productions were reduced by 89.6 and 75.6%, respectively at 4 and 8 μg/mL of AgNPs, suggesting anti-quorum sensing activity. Exopolysaccharide production was decreased by 61-79 and 84% for Gram -ve and Gram +ve pathogens respectively. Flagellar driven swarming mobility was also reduced significantly. Furthermore, In vivo study confirmed their tolerability in mice, indicating their clinical perspective. Collective, we claim that the synthesized AgNPs have anti-metastasis as well as anti-microbial activities. Hence, this can be further tested for therapeutic options to treat cancer and secondary bacterial infections.
Keywords: anti-biofilm; anti-metastasis; molecular docking; quorum sensing; silver nanoparticles (AgNPs).
Figures



References
-
- Accelrys Software Inc. (2013). Discovery Studio Modeling Environment, Release 4.0. San Diego, CA: Author.
-
- Ahluwalia V. K., Kidwai M. (2004). “Microwave induced green synthesis,” in New Trends in Green Chemistry, eds Ahluwalia V. K., Kidwai M. (Dordrecht: Springer; ), 59–72. 10.1007/978-1-4020-3175-5_9 - DOI
-
- Ahmed F., Kumar S., Arshi N., Anwar M. S., Koo B. H., Lee C. G. (2011). Rapid and cost effective synthesis of ZnO nanorods using microwave irradiation technique. Funct. Mater. Lett. 4 1–5. 10.1142/S1793604711001531 - DOI
-
- Alajmi M. F., Ahmed J., Hussain A., Ahamad T., Alhokbany N., Amir S., et al. (2018). Green synthesis of ultrafine Fe3O4 nanoparticles using aqueous extracts of plant leaves for efficient bifunctional electro-catalytic activity. Appl. Nanosci. 8 1427–1435. 10.1007/s13204-018-0795-8 - DOI
LinkOut - more resources
Full Text Sources

