The glycosylation landscape of pancreatic cancer
- PMID: 30854032
- PMCID: PMC6388511
- DOI: 10.3892/ol.2019.9885
The glycosylation landscape of pancreatic cancer
Abstract
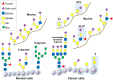
Pancreatic adenocarcinoma is a lethal disease with a 5-year survival rate of <5%, the lowest of all types of cancer. The diagnosis of pancreatic cancer relies on imaging and tissue biopsy, and the only curative therapy is complete surgical resection. Pancreatic cancer has the propensity to metastasise at an early stage and the majority of patients are diagnosed when surgery is no longer an option. Hence, there is an urgent need to identify biomarkers to enable early diagnosis, and to develop new therapeutic strategies. One approach for this involves targeting cancer-associated glycans. The most widely used serological marker in pancreatic cancer is the carbohydrate antigen CA 19-9 which contains a glycan known as sialyl Lewis A (sLeA). The CA 19-9 assay is used routinely to monitor response to treatment, but concerns have been raised about its sensitivity and specificity as a diagnostic biomarker. In addition to sLeA, a wide range of alterations to other important glycans have been observed in pancreatic cancer. These include increases in the sialyl Lewis X antigen (sLex), an increase in truncated O-glycans (Tn and sTn), increased branched and fucosylated N-glycans, upregulation of specific proteoglycans and galectins, and increased O-GlcNAcylation. Growing evidence supports crucial roles for glycans in all stages of cancer progression, and it is well established that glycans regulate tumour proliferation, invasion and metastasis. The present review describes the biological significance of glycans in pancreatic cancer, and discusses the clinical value of exploiting aberrant glycosylation to improve the diagnosis and treatment of this deadly disease.
Keywords: biomarkers; glycans; glycosylation; pancreatic cancer; treatment.
Figures
References
-
- Klöppel G, Adsay NV. Chronic pancreatitis and the differential diagnosis versus pancreatic cancer. Arch Pathol Lab Med. 2009;133:382–387. - PubMed
-
- Meezan E, Wu HC, Black PH, Robbins PW. Comparative studies on the carbohydrate-containing membrane components of normal and virus-transformed mouse fibroblasts. II. Separation of glycoproteins and glycopeptides by sephadex chromatography. Biochemistry. 1969;8:2518–2524. doi: 10.1021/bi00834a039. - DOI - PubMed
Publication types
LinkOut - more resources
Full Text Sources
Other Literature Sources
