Immunopathological characterization of ovarian teratomas associated with anti-N-methyl-D-aspartate receptor encephalitis
- PMID: 30857565
- PMCID: PMC6410529
- DOI: 10.1186/s40478-019-0693-7
Immunopathological characterization of ovarian teratomas associated with anti-N-methyl-D-aspartate receptor encephalitis
Abstract
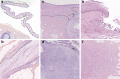
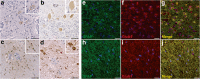
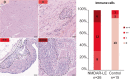
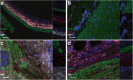
Encephalitis with anti-NMDAR antibodies (NMDAR-E) is a severe autoimmune neurological disorder, defined by a clinical presentation of encephalitis and the presence of IgG targeting the GluN1 subunit of NMDA receptors in the CSF. An underlying ovarian teratoma is commonly associated with this autoimmune disease suggesting a role of the tumor in immunopathogenesis. In this study, we characterized the salient histopathological features of 27 ovarian teratomas associated with NMDAR-E (3 immature and 24 mature teratomas) and 40 controls without associated encephalitis. All but one NMDAR-E-associated teratomas contained a nervous tissue component, while less than 40% of control teratomas did (p < 0.001). GluN1 expression by teratomatous nervous tissue seemed to be more often glial in NMDAR-E teratomas than in control teratomas (73% vs. 29%, p < 0.05). Strikingly, 3 out of 24 NMDAR-E-associated mature teratomas contained neuroglial tissue exhibiting histopathological features of central nervous system neuroglial tumor, while such glioma-like features are exceptionally described in the literature on ovarian teratomas. Moreover, NMDAR-E associated teratomas differed from sporadic ovarian teratomas by consistent and prominent infiltration of the nervous tissue component by immune cells, comprised of T- and B-cells and mature dendritic cells organized in tertiary lymphoid structures, with IgG and IgA deposits and plasma cells in close contact to the neuroglial tissue.These data demonstrate an association between massive infiltration of NMDAR-E-associated teratomas by immune cells and particular glial features of its neuroglial component, suggesting that this glial tissue might be involved in triggering or sustaining the anti-tumor response associated with the auto-immune neurological disease.
Keywords: Anti-NMDAR encephalitis; Autoimmunity; Ovarian teratoma.
Conflict of interest statement
Ethics approval and consent to participate
Written consent was obtained from all patients, and this study was approved by the institutional review board of the University Claude Bernard Lyon 1 and Hospices Civils de Lyon.
Competing interests
The authors have declared that they have no competing interests.
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Figures


References
-
- Bost C, Chanson E, Picard G, Meyronet D, Mayeur M-E, Ducray F, Rogemond V, Psimaras D, Antoine J-C, Delattre J-Y, Desestret V, Honnorat J (2018) Malignant tumors in autoimmune encephalitis with anti-NMDA receptor antibodies. J Neurol. 10.1007/s00415-018-8970-0 - PubMed
Publication types
MeSH terms
Supplementary concepts
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Miscellaneous

