A multicellular way of life for a multipartite virus
- PMID: 30857590
- PMCID: PMC6414197
- DOI: 10.7554/eLife.43599
A multicellular way of life for a multipartite virus
Abstract
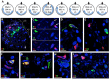
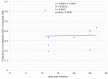
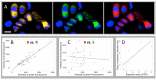
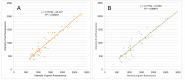
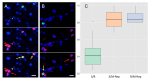
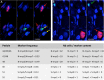
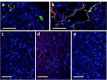
A founding paradigm in virology is that the spatial unit of the viral replication cycle is an individual cell. Multipartite viruses have a segmented genome where each segment is encapsidated separately. In this situation the viral genome is not recapitulated in a single virus particle but in the viral population. How multipartite viruses manage to efficiently infect individual cells with all segments, thus with the whole genome information, is a long-standing but perhaps deceptive mystery. By localizing and quantifying the genome segments of a nanovirus in host plant tissues we show that they rarely co-occur within individual cells. We further demonstrate that distinct segments accumulate independently in different cells and that the viral system is functional through complementation across cells. Our observation deviates from the classical conceptual framework in virology and opens an alternative possibility (at least for nanoviruses) where the infection can operate at a level above the individual cell level, defining a viral multicellular way of life.
Keywords: evolution; genome; infectious disease; microbiology; multipartite virus; plant; population genetics; virus; virus-host interaction.
© 2019, Sicard et al.
Conflict of interest statement
AS, EP, RG, MV, MY, CU, MP, SG, YM, SB No competing interests declared
Figures

References
-
- Brakke MK, Black LM, Wyckoff RWG. The sedimentation rate of potato yellow-dwarf virus. American Journal of Botany. 1951;38:332–342. doi: 10.1002/j.1537-2197.1951.tb14832.x. - DOI
Publication types
MeSH terms
Substances
Associated data
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

