HMGB1-RAGE Signaling Plays a Role in Organic Dust-Induced Microglial Activation and Neuroinflammation
- PMID: 30859215
- PMCID: PMC6542342
- DOI: 10.1093/toxsci/kfz071
HMGB1-RAGE Signaling Plays a Role in Organic Dust-Induced Microglial Activation and Neuroinflammation
Abstract
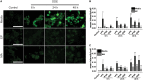
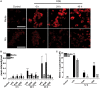
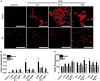
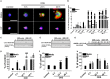
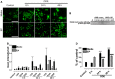
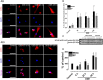
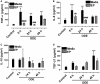
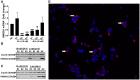
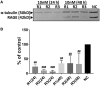
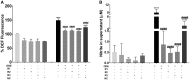
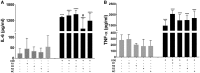
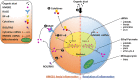
Occupational exposure to contaminants in agriculture and other industries is known to cause significant respiratory ailments. The effect of organic dust on lung inflammation and tissue remodeling has been actively investigated over many years but the adverse effect of organic dust-exposure on the central vital organ brain is beginning to emerge. Brain microglial cells are a major driver of neuroinflammation upon exposure to danger signals. Therefore, we tested a hypothesis that organic dust-exposure of microglial cells induces microglial cell activation and inflammation through HMGB1-RAGE signaling. Mouse microglial cells were exposed to organic dust extract showed a time-dependent increase in cytoplasmic translocation of high-mobility group box 1 (HMGB1) from the nucleus, increased expression of receptor for advanced glycation end products (RAGE) and activation of Iba1 as compared to control cells. Organic dust also induced reactive oxygen species generation, NF-κB activation, and proinflammatory cytokine release. To establish a functional relevance of HMGB1-RAGE activation in microglia-mediated neuroinflammation, we used both pharmacological and genetic approaches involving HMGB1 translocation inhibitor ethyl pyruvate (EP), anti-HMGB1 siRNA, and NOX-inhibitor mitoapocynin. Interestingly, EP effectively reduced HMGB1 nucleocytoplasmic translocation and RAGE expression along with reactive oxygen species (ROS) generation and TNF-α and IL-6 production but not NF-κB activation. HMGB1 knockdown by siRNA also reduced both ROS and reactive nitrogen species (RNS) and IL-6 levels but not TNF-α. NOX2 inhibitor mitoapocynin significantly reduced RNS levels. Collectively, our results demonstrate that organic dust activates HMGB1-RAGE signaling axis to induce a neuroinflammatory response in microglia and that attenuation of HMGB1-RAGE activation by EP and mitoapocynin treatments or genetic knockdown can dampen the neuroinflammation.
Keywords: HMGB1; RAGE; microglia; neuroinflammation; organic dust.
© The Author(s) 2019. Published by Oxford University Press on behalf of the Society of Toxicology. All rights reserved. For permissions, please e-mail: journals.permissions@oup.com.
Figures
Similar articles
-
Ethyl pyruvate reduces organic dust-induced airway inflammation by targeting HMGB1-RAGE signaling.Respir Res. 2019 Feb 6;20(1):27. doi: 10.1186/s12931-019-0992-3. Respir Res. 2019. PMID: 30728013 Free PMC article.
-
Omega-3 polyunsaturated fatty acid supplementation attenuates microglial-induced inflammation by inhibiting the HMGB1/TLR4/NF-κB pathway following experimental traumatic brain injury.J Neuroinflammation. 2017 Jul 24;14(1):143. doi: 10.1186/s12974-017-0917-3. J Neuroinflammation. 2017. PMID: 28738820 Free PMC article.
-
HMGB1/RAGE axis mediates stress-induced RVLM neuroinflammation in mice via impairing mitophagy flux in microglia.J Neuroinflammation. 2020 Jan 10;17(1):15. doi: 10.1186/s12974-019-1673-3. J Neuroinflammation. 2020. PMID: 31924219 Free PMC article.
-
Every Cloud Has a Silver Lining: Proneurogenic Effects of Aβ Oligomers and HMGB-1 via Activation of the RAGE-NF-κB Axis.CNS Neurol Disord Drug Targets. 2017;16(10):1066-1079. doi: 10.2174/1871527315666160803153459. CNS Neurol Disord Drug Targets. 2017. PMID: 27488419 Review.
-
Convergence and amplification of toll-like receptor (TLR) and receptor for advanced glycation end products (RAGE) signaling pathways via high mobility group B1 (HMGB1).Angiogenesis. 2008;11(1):91-9. doi: 10.1007/s10456-008-9093-5. Epub 2008 Feb 9. Angiogenesis. 2008. PMID: 18264787 Review.
Cited by
-
The pivotal role of inflammatory factors in glaucoma: a systematic review.Front Immunol. 2025 May 23;16:1577200. doi: 10.3389/fimmu.2025.1577200. eCollection 2025. Front Immunol. 2025. PMID: 40486511 Free PMC article.
-
Role of high mobility group box protein 1 in depression: A mechanistic and therapeutic perspective.World J Psychiatry. 2022 Jun 19;12(6):779-786. doi: 10.5498/wjp.v12.i6.779. eCollection 2022 Jun 19. World J Psychiatry. 2022. PMID: 35978968 Free PMC article. Review.
-
Ethyl pyruvate, a versatile protector in inflammation and autoimmunity.Inflamm Res. 2022 Feb;71(2):169-182. doi: 10.1007/s00011-021-01529-z. Epub 2022 Jan 9. Inflamm Res. 2022. PMID: 34999919 Free PMC article. Review.
-
Pre-exposure to hydrogen sulfide modulates the innate inflammatory response to organic dust.Cell Tissue Res. 2021 Apr;384(1):129-148. doi: 10.1007/s00441-020-03333-3. Epub 2021 Jan 6. Cell Tissue Res. 2021. PMID: 33409657 Free PMC article.
-
Is Oxytocin "Nature's Medicine"?Pharmacol Rev. 2020 Oct;72(4):829-861. doi: 10.1124/pr.120.019398. Pharmacol Rev. 2020. PMID: 32912963 Free PMC article. Review.
References
-
- Charavaryamath C., Juneau V., Suri S. S., Janardhan K. S., Townsend H., Singh B. (2008). Role of toll-like receptor 4 in lung inflammation following exposure to swine barn air. Exp. Lung Res. 34, 19–35. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

