Identification and characterization of potent and selective aquaporin-3 and aquaporin-7 inhibitors
- PMID: 30862673
- PMCID: PMC6509502
- DOI: 10.1074/jbc.RA118.006083
Identification and characterization of potent and selective aquaporin-3 and aquaporin-7 inhibitors
Abstract
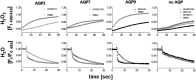
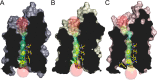
The aquaglyceroporins are a subfamily of aquaporins that conduct both water and glycerol. Aquaporin-3 (AQP3) has an important physiological function in renal water reabsorption, and AQP3-mediated hydrogen peroxide (H2O2) permeability can enhance cytokine signaling in several cell types. The related aquaglyceroporin AQP7 is required for dendritic cell chemokine responses and antigen uptake. Selective small-molecule inhibitors are desirable tools for investigating the biological and pathological roles of these and other AQP isoforms. Here, using a calcein fluorescence quenching assay, we screened a library of 7360 drug-like small molecules for inhibition of mouse AQP3 water permeability. Hit confirmation and expansion with commercially available substances identified the ortho-chloride-containing compound DFP00173, which inhibited mouse and human AQP3 with an IC50 of ∼0.1-0.4 μm but had low efficacy toward mouse AQP7 and AQP9. Surprisingly, inhibitor specificity testing revealed that the methylurea-linked compound Z433927330, a partial AQP3 inhibitor (IC50, ∼0.7-0.9 μm), is a potent and efficacious inhibitor of mouse AQP7 water permeability (IC50, ∼0.2 μm). Stopped-flow light scattering measurements confirmed that DFP00173 and Z433927330 inhibit AQP3 glycerol permeability in human erythrocytes. Moreover, DFP00173, Z433927330, and the previously identified AQP9 inhibitor RF03176 blocked aquaglyceroporin H2O2 permeability. Molecular docking to AQP3, AQP7, and AQP9 homology models suggested interactions between these inhibitors and aquaglyceroporins at similar binding sites. DFP00173 and Z433927330 constitute selective and potent AQP3 and AQP7 inhibitors, respectively, and contribute to a set of isoform-specific aquaglyceroporin inhibitors that will facilitate the evaluation of these AQP isoforms as drug targets.
Keywords: CHO cell assay; HyPer; aquaporin; aquaporin inhibitor; calcein; glycerol; hydrogen peroxide; inhibitor; molecular docking; residual pore.
© 2019 Sonntag et al.
Conflict of interest statement
The authors declare that they have no conflicts of interest with the contents of this article
Figures




References
-
- Ishibashi K., Sasaki S., Fushimi K., Uchida S., Kuwahara M., Saito H., Furukawa T., Nakajima K., Yamaguchi Y., and Gojobori T. (1994) Molecular cloning and expression of a member of the aquaporin family with permeability to glycerol and urea in addition to water expressed at the basolateral membrane of kidney collecting duct cells. Proc. Natl. Acad. Sci. U.S.A. 91, 6269–6273 10.1073/pnas.91.14.6269 - DOI - PMC - PubMed
-
- Ecelbarger C. A., Terris J., Frindt G., Echevarria M., Marples D., Nielsen S., and Knepper M. A. (1995) Aquaporin-3 water channel localization and regulation in rat kidney. Am. J. Physiol. 269, F663–F672 - PubMed
Publication types
MeSH terms
Substances
Associated data
- Actions
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

