First-line tyrosine kinase inhibitors in EGFR mutation-positive non-small-cell lung cancer: a network meta-analysis
- PMID: 30863108
- PMCID: PMC6388947
- DOI: 10.2147/OTT.S189438
First-line tyrosine kinase inhibitors in EGFR mutation-positive non-small-cell lung cancer: a network meta-analysis
Abstract
Background: EGFR-tyrosine kinase inhibitors (EGFR-TKIs) including afatinib, dacomitinib, erlotinib, gefitinib, and osimertinib have proven efficacy in terms of progression-free survival (PFS) in patients with non-small-cell lung cancer (NSCLC) harboring EGFR mutations. However, an overall view for comparing efficacy and toxicity on a meta-level is lacking. This study compared efficacy and toxicity of first-line treatment with five different EGFR-TKIs by conducting a network meta-analysis (NMA).
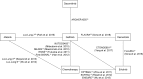
Methods: A systematic review was performed, aiming to find eligible literature. Data of PFS, overall survival (OS), objective response rate (ORR), and adverse events were extracted. An NMA based on Bayesian statistics was established to synthesize the efficacy and toxicity of all treatments.
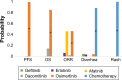
Results: Thirteen randomized controlled trials, including data from 3,539 patients with EGFR-mutated NSCLC, were analyzed. Rank probabilities showed that osimertinib had a potentially better efficacy in terms of PFS and OS compared to all other TKIs. For ORR, afatinib and osimertinib showed a trend of superiority compared to the other four TKIs. Furthermore, there was a high risk of diarrhea and rash for patients treated with afatinib or dacomitinib as well as a moderate risk for treatment with erlotinib, gefitinib, and osimertinib.
Conclusion: Our study showed a favorable efficacy of osimertinib in terms of PFS and OS compared to all other EGFR-TKIs in patients with NSCLC harboring activating EGFR mutations. Furthermore, gefitinib, erlotinib, and osimertinib were associated with fewer toxicities compared to the other TKIs. Therefore, osimertinib is indicated as a preferable first-line TKI in patients with activating EGFR-mutated NSCLC.
Keywords: EGFR-TKI; afatinib; erlotinib; gefitinib; network meta-analysis; osimertinib.
Conflict of interest statement
Disclosure Carin A Uyl-de Groot reports grants received from Boehringer-Ingelheim, Janssen-Cilag B.V., Genzyme, Astellas Pharma B.V., Sanofi Netherlands, Roche Netherlands B.V., AstraZeneca Netherlands, Amgen Europe B.V., Gilead Sciences, Merck, and Bayer, outside the submitted work. The other authors report no conflicts of interest in this work.
Figures

References
-
- World Health Organization Fact sheet cancer. [Accessed September, 2017]. Available from: http://www.who.int/mediacentre/factsheets/fs297/en/
-
- Alvarez M, Roman E, Santos ES, Raez LE. New targets for non-small-cell lung cancer therapy. Expert Rev Anticancer Ther. 2007;7(10):1423–1437. - PubMed
Publication types
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

