Rosiglitazone, a Ligand to PPAR γ, Improves Blood Pressure and Vascular Function through Renin-Angiotensin System Regulation
- PMID: 30863432
- PMCID: PMC6378057
- DOI: 10.1155/2019/1371758
Rosiglitazone, a Ligand to PPAR γ, Improves Blood Pressure and Vascular Function through Renin-Angiotensin System Regulation
Abstract
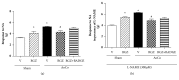
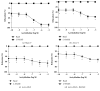
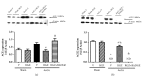
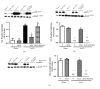
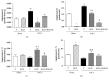
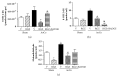
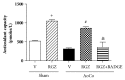
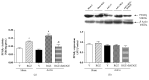
Rosiglitazone (RGZ), a peroxisome proliferator-activated receptor gamma (PPARγ) ligand, has been reported to act as insulin sensitizer and exert cardiovascular actions. In this work, we hypothesized that RGZ exerts a PPARγ-dependent regulation of blood pressure through modulation of angiotensin-converting enzyme (ACE)-type 2 (ACE2)/angiotensin-(1-7)/angiotensin II type-2 receptor (AT2R) axis in an experimental model of high blood pressure. We carried on experiments in normotensive (Sham) and aortic coarctation (AoCo)-induced hypertensive male Wistar rats. Both sham and AoCo rats were treated 7 days with vehicle (V), RGZ (5 mg/kg/day), or RGZ+BADGE (120 mg/kg/day) post-coarctation. We measured blood pressure and vascular reactivity on aortic rings, as well as the expression of renin-angiotensin system (RAS) proteins. We found that RGZ treatment in AoCo group decreases blood pressure values and improves vascular response to acetylcholine, both parameters dependent on PPARγ-stimulation. RGZ lowered serum angiotensin II (AngII) but increased Ang-(1-7) levels. It also decreased 8-hydroxy-2'-deoxyguanosine (8-OH-2dG), malondialdehyde (MDA), and improved the antioxidant capacity. Regarding protein expression of RAS, RGZ decreases ACE and angiotensin II type 1 receptor (AT1R) and improved ACE2, AT2R, and Mas receptor in AoCo rats. Additionally, an in silico analysis revealed that 5'UTR regions of RAS and PPARγ share motifs with a transcriptional regulatory role. We conclude that RGZ lowers blood pressure values by increasing the expression of RAS axis proteins ACE2 and AT2R, decreasing the levels of AngII and increasing levels of Ang-(1-7) in a PPARγ-dependent manner. The in silico analysis is a valuable tool to predict the interaction between PPARγ and RAS.
Figures

Similar articles
-
Effect of clofibrate on vascular reactivity in a model of high blood pressure secondary to aortic coarctation.Pharmacol Rep. 2010 Sep-Oct;62(5):874-82. doi: 10.1016/s1734-1140(10)70347-9. Pharmacol Rep. 2010. PMID: 21098870
-
PPARalpha stimulation exerts a blood pressure lowering effect through different mechanisms in a time-dependent manner.Eur J Pharmacol. 2010 Feb 10;627(1-3):185-93. doi: 10.1016/j.ejphar.2009.10.039. Epub 2009 Oct 24. Eur J Pharmacol. 2010. PMID: 19857485
-
Renin-Angiotensin System Responds to Prolonged Hypotensive Effect Induced by Mandibular Extension in Spontaneously Hypertensive Rats.Front Physiol. 2018 Nov 15;9:1613. doi: 10.3389/fphys.2018.01613. eCollection 2018. Front Physiol. 2018. PMID: 30498455 Free PMC article.
-
ACE2 and vasoactive peptides: novel players in cardiovascular/renal remodeling and hypertension.Ther Adv Cardiovasc Dis. 2015 Aug;9(4):217-37. doi: 10.1177/1753944715597623. Epub 2015 Aug 13. Ther Adv Cardiovasc Dis. 2015. PMID: 26275770 Review.
-
The vasoprotective axes of the renin-angiotensin system: Physiological relevance and therapeutic implications in cardiovascular, hypertensive and kidney diseases.Pharmacol Res. 2017 Nov;125(Pt A):21-38. doi: 10.1016/j.phrs.2017.06.005. Epub 2017 Jun 12. Pharmacol Res. 2017. PMID: 28619367 Free PMC article. Review.
Cited by
-
Obesity, Diabetes Mellitus, and Metabolic Syndrome: Review in the Era of COVID-19.Clin Nutr Res. 2022 Oct 24;11(4):331-346. doi: 10.7762/cnr.2022.11.4.331. eCollection 2022 Oct. Clin Nutr Res. 2022. PMID: 36381471 Free PMC article. Review.
-
SARS-CoV-2, ACE2, and Hydroxychloroquine: Cardiovascular Complications, Therapeutics, and Clinical Readouts in the Current Settings.Pathogens. 2020 Jul 7;9(7):546. doi: 10.3390/pathogens9070546. Pathogens. 2020. PMID: 32645974 Free PMC article. Review.
-
PPAR Gamma: From Definition to Molecular Targets and Therapy of Lung Diseases.Int J Mol Sci. 2021 Jan 15;22(2):805. doi: 10.3390/ijms22020805. Int J Mol Sci. 2021. PMID: 33467433 Free PMC article. Review.
-
Metabolic Syndrome and COVID 19: Endocrine-Immune-Vascular Interactions Shapes Clinical Course.Endocrinology. 2020 Oct 1;161(10):bqaa112. doi: 10.1210/endocr/bqaa112. Endocrinology. 2020. PMID: 32603424 Free PMC article. Review.
-
COVID-19: is there a link between the course of infection and pharmacological agents in diabetes?J Endocrinol Invest. 2020 Aug;43(8):1053-1060. doi: 10.1007/s40618-020-01318-1. Epub 2020 Jun 3. J Endocrinol Invest. 2020. PMID: 32495299 Free PMC article. Review.
References
-
- Wilson J. L., Duan R., El-Marakby A., Alhashim A., Lee D. L. Peroxisome proliferator activated receptor- agonist slows the progression of hypertension, attenuates plasma interleukin-6 levels and renal inflammatory markers in angiotensin II infused mice. PPAR Research. 2012;2012:7. doi: 10.1155/2012/645969.645969 - DOI - PMC - PubMed
LinkOut - more resources
Full Text Sources
Miscellaneous

