True 99th centile of high sensitivity cardiac troponin for hospital patients: prospective, observational cohort study
- PMID: 30867154
- PMCID: PMC6415647
- DOI: 10.1136/bmj.l729
True 99th centile of high sensitivity cardiac troponin for hospital patients: prospective, observational cohort study
Abstract
Objective: To determine the distribution, and specifically the true 99th centile, of high sensitivity cardiac troponin I (hs-cTnI) for a whole hospital population by applying the hs-cTnI assay currently used routinely at a large teaching hospital.
Design: Prospective, observational cohort study.
Setting: University Hospital Southampton NHS Foundation Trust, Southampton, United Kingdom, between 29 June 2017 and 24 August 2017.
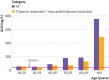
Participants: 20 000 consecutive inpatients and outpatients undergoing blood tests for any clinical reason. Hs-cTnI concentrations were measured in all study participants and nested for analysis except when the supervising doctor had requested hs-cTnI for clinical reasons.
Main outcome measures: Distribution of hs-cTnI concentrations of all study participants and specifically the 99th centile.
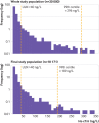
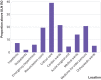
Results: The 99th centile of hs-cTnI for the whole population was 296 ng/L compared with the manufacturer's quoted level of 40 ng/L (currently used clinically as the upper limit of normal; ULN). Hs-cTnI concentrations were greater than 40 ng/L in one in 20 (5.4%, n=1080) of the total population. After excluding participants diagnosed as having acute myocardial infarction (n=122) and those in whom hs-cTnI was requested for clinical reasons (n=1707), the 99th centile was 189 ng/L for the remainder (n=18 171). The 99th centile was 563 ng/L for inpatients (n=4759) and 65 ng/L for outpatients (n=9280). Patients from the emergency department (n=3706) had a 99th centile of 215 ng/L, with 6.07% (n=225) greater than the recommended ULN. 39.02% (n=48) of all patients from the critical care units (n=123) and 14.16% (n=67) of all medical inpatients had an hs-cTnI concentration greater than the recommended ULN.
Conclusions: Of 20 000 consecutive patients undergoing a blood test for any clinical reason at our hospital, one in 20 had an hs-cTnI greater than the recommended ULN. These data highlight the need for clinical staff to interpret hs-cTnI concentrations carefully, particularly when applying the recommended ULN to diagnose acute myocardial infarction, in order to avoid misdiagnosis in the absence of an appropriate clinical presentation.
Trial registration: Clinicaltrials.gov NCT03047785.
Published by the BMJ Publishing Group Limited. For permission to use (where not already granted under a licence) please go to http://group.bmj.com/group/rights-licensing/permissions.
Conflict of interest statement
Competing interests: All authors have completed the ICMJE uniform disclosure form at www.icmje.org/coi_disclosure.pdf and declare: support from Beckman Coulter for the submitted work; NC received unrestricted research grants from Boston Scientific, Haemonetics, Heartflow, Beckmann Coulter; speaker fees or consultancy fees from Haemonetics, Abbot Vascular, Heartflow, and Boston Scientific; and travel sponsorship from Biosensors, Abbot, Lilly/D-S, St Jude Medical, and Medtronic. Ethical approval: This research project was undertaken according to the principles of Good Clinical Practice and the Declaration of Helsinki. The study was approved by the local ethical committee who then referred it to the Health Research Authority UK and its independent Confidentiality Advisory Group for further approval (REC reference: 17/SC/0042; IRAS project ID: 215262).
Figures
Similar articles
-
No increase in the incidence of cardiac troponin I concentration above the 99th percentile by Siemens Centaur high-sensitivity compared to the contemporary assay.Clin Biochem. 2021 Mar;89:77-80. doi: 10.1016/j.clinbiochem.2020.12.001. Epub 2020 Dec 6. Clin Biochem. 2021. PMID: 33296707
-
High-sensitivity troponin in the evaluation of patients with suspected acute coronary syndrome: a stepped-wedge, cluster-randomised controlled trial.Lancet. 2018 Sep 15;392(10151):919-928. doi: 10.1016/S0140-6736(18)31923-8. Epub 2018 Aug 28. Lancet. 2018. PMID: 30170853 Free PMC article. Clinical Trial.
-
Single test rule-out of acute myocardial infarction using the limit of detection of a new high-sensitivity troponin I assay.Clin Biochem. 2020 Apr;78:4-9. doi: 10.1016/j.clinbiochem.2020.02.014. Epub 2020 Mar 2. Clin Biochem. 2020. PMID: 32135083
-
High-sensitivity cardiac troponin assays: From improved analytical performance to enhanced risk stratification.Crit Rev Clin Lab Sci. 2017 May;54(3):143-172. doi: 10.1080/10408363.2017.1285268. Epub 2017 May 1. Crit Rev Clin Lab Sci. 2017. PMID: 28457177 Review.
-
Short-term biological variation and diurnal rhythm of cardiac troponin I (Access hs-TnI) in healthy subjects.Clin Chim Acta. 2020 May;504:163-167. doi: 10.1016/j.cca.2020.02.004. Epub 2020 Feb 5. Clin Chim Acta. 2020. PMID: 32035091 Review.
Cited by
-
A pipeline to further enhance quality, integrity and reusability of the NCCID clinical data.Sci Data. 2023 Jul 27;10(1):493. doi: 10.1038/s41597-023-02340-7. Sci Data. 2023. PMID: 37500661 Free PMC article.
-
Acute Myocardial Infarction in the Setting of Pulmonary Hypertension due to a Patent Foramen Ovale and Paradoxical Embolism.Case Rep Pulmonol. 2024 Jul 18;2024:6725308. doi: 10.1155/2024/6725308. eCollection 2024. Case Rep Pulmonol. 2024. PMID: 39055527 Free PMC article.
-
High-Sensitivity Troponin I and Cardiovascular Events in Stable Coronary Artery Disease: Insights from a Longitudinal Outpatient Study.Int J Mol Sci. 2023 Dec 9;24(24):17286. doi: 10.3390/ijms242417286. Int J Mol Sci. 2023. PMID: 38139114 Free PMC article.
-
Distribution of contemporary sensitivity troponin in the emergency department and relationship to 30-day mortality: The CHARIOT-ED substudy.Clin Med (Lond). 2020 Nov;20(6):528-534. doi: 10.7861/clinmed.2020-0267. Clin Med (Lond). 2020. PMID: 33199315 Free PMC article.
-
Cardiac-specific troponin-I (cTnI) in a post-mortem setting.Int J Legal Med. 2025 Sep;139(5):2539-2548. doi: 10.1007/s00414-025-03526-x. Epub 2025 May 23. Int J Legal Med. 2025. PMID: 40407876 Free PMC article.
References
-
- Thygesen K, Alpert JS, Jaffe AS, et al. Joint ESC/ACCF/AHA/WHF Task Force for Universal Definition of Myocardial Infarction. Authors/Task Force Members Chairpersons. Biomarker Subcommittee. ECG Subcommittee. Imaging Subcommittee. Classification Subcommittee. Intervention Subcommittee. Trials & Registries Subcommittee. Trials & Registries Subcommittee. Trials & Registries Subcommittee. Trials & Registries Subcommittee. ESC Committee for Practice Guidelines (CPG) Document Reviewers Third universal definition of myocardial infarction. J Am Coll Cardiol 2012;60:1581-98. 10.1016/j.jacc.2012.08.001 - DOI - PubMed
-
- Thygesen K, Alpert JS, Jaffe AS, et al. Executive Group on behalf of the Joint European Society of Cardiology (ESC)/American College of Cardiology (ACC)/American Heart Association (AHA)/World Heart Federation (WHF) Task Force for the Universal Definition of Myocardial Infarction Fourth Universal Definition of Myocardial Infarction (2018). J Am Coll Cardiol 2018;72:2231-64. 10.1016/j.jacc.2018.08.1038 - DOI - PubMed
Publication types
MeSH terms
Substances
Associated data
LinkOut - more resources
Full Text Sources
Medical
Research Materials
Miscellaneous
