Efficacy of MEK inhibition in patients with histiocytic neoplasms
- PMID: 30867592
- PMCID: PMC6438729
- DOI: 10.1038/s41586-019-1012-y
Efficacy of MEK inhibition in patients with histiocytic neoplasms
Abstract
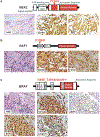
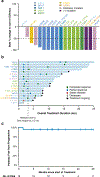
Histiocytic neoplasms are a heterogeneous group of clonal haematopoietic disorders that are marked by diverse mutations in the mitogen-activated protein kinase (MAPK) pathway1,2. For the 50% of patients with histiocytosis who have BRAFV600 mutations3-5, RAF inhibition is highly efficacious and has markedly altered the natural history of the disease6,7. However, no standard therapy exists for the remaining 50% of patients who lack BRAFV600 mutations. Although ERK dependence has been hypothesized to be a consistent feature across histiocytic neoplasms, this remains clinically unproven and many of the kinase mutations that are found in patients who lack BRAFV600 mutations have not previously been biologically characterized. Here we show ERK dependency in histiocytoses through a proof-of-concept clinical trial of cobimetinib, an oral inhibitor of MEK1 and MEK2, in patients with histiocytoses. Patients were enrolled regardless of their tumour genotype. In parallel, MAPK alterations that were identified in treated patients were characterized for their ability to activate ERK. In the 18 patients that we treated, the overall response rate was 89% (90% confidence interval of 73-100). Responses were durable, with no acquired resistance to date. At one year, 100% of responses were ongoing and 94% of patients remained progression-free. Cobimetinib treatment was efficacious regardless of genotype, and responses were observed in patients with ARAF, BRAF, RAF1, NRAS, KRAS, MEK1 (also known as MAP2K1) and MEK2 (also known as MAP2K2) mutations. Consistent with the observed responses, the characterization of the mutations that we identified in these patients confirmed that the MAPK-pathway mutations were activating. Collectively, these data demonstrate that histiocytic neoplasms are characterized by a notable dependence on MAPK signalling-and that they are consequently responsive to MEK inhibition. These results extend the benefits of molecularly targeted therapy to the entire spectrum of patients with histiocytosis.
Figures


References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases
Research Materials
Miscellaneous

