Cost effectiveness and affordability of trastuzumab in sub-Saharan Africa for early stage HER2-positive breast cancer
- PMID: 30867655
- PMCID: PMC6396469
- DOI: 10.1186/s12962-019-0174-7
Cost effectiveness and affordability of trastuzumab in sub-Saharan Africa for early stage HER2-positive breast cancer
Abstract
Background: Breast cancer is the second most common cancer worldwide, the most common among women, and the most frequent cause of death among women in less developed regions. Trastuzumab is a humanized monoclonal antibody that downregulates the extracellular domain of the HER2 protein. Using trastuzumab to treat women with localized HER2-positive breast cancer has been shown to improve survival. The objective of this study is to explore the cost-effectiveness of adjuvant trastuzumab, from a societal perspective, in 11 African countries. In addition, we aimed to establish value-based prices for trastuzumab based on the gross domestic product per capita in each country.
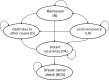
Methods: We developed a Markov model in order to assess the costs and benefits associated with trastuzumab treatment over a lifetime horizon. A probabilistic sensitivity analysis was performed in order to estimate the impact of uncertainty of parameter-values on the results. Efficacy inputs were derived using clinical trial data from non-African countries.
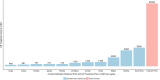
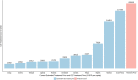
Results: In the base case analysis, trastuzumab yielded a gain ranging from 0.92 LYs in Nigeria to 1.07 LYs in South Africa, and 0.9 QALYs in Nigeria to 1.02 QALYs in South Africa. The incremental cost ranged from 19,561 USD in Nigeria to 19,997 USD in Congo, and an incremental cost-effectiveness ratio ranging from 19,534 USD/QALY in South Africa to 21,697 USD/QALY in Nigeria. Using willingness to pay estimates based on World Health Organization recommendations, trastuzumab appear to not be cost-effective in all countries analyzed. Cost-effectiveness estimates were most sensitive to the discount rate, trastuzumab cost, and the hazard ratio.
Conclusions: Trastuzumab does not appear to be cost effective in the African countries analyzed. In order for trastuzumab to be cost-effective, the costs of treatment would require significant discounts.
Keywords: Cost-effectiveness; Sub-Saharan Africa; Trastuzumab.
Figures
References
-
- Institute of Medicine. Cancer control opportunities in low- and middle-income countries. vol 57. 2007. https://www.ncbi.nlm.nih.gov/books/NBK54030/pdf/Bookshelf_NBK54030.pdf. Accessed Sept 14 2017.
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous