Hydroxycobalamin Reveals the Involvement of Hydrogen Sulfide in the Hypoxic Responses of Rat Carotid Body Chemoreceptor Cells
- PMID: 30871199
- PMCID: PMC6466535
- DOI: 10.3390/antiox8030062
Hydroxycobalamin Reveals the Involvement of Hydrogen Sulfide in the Hypoxic Responses of Rat Carotid Body Chemoreceptor Cells
Abstract
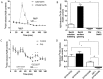
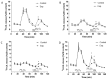
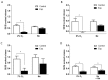
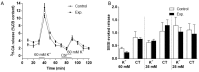
Carotid body (CB) chemoreceptor cells sense arterial blood PO₂, generating a neurosecretory response proportional to the intensity of hypoxia. Hydrogen sulfide (H₂S) is a physiological gaseous messenger that is proposed to act as an oxygen sensor in CBs, although this concept remains controversial. In the present study we have used the H₂S scavenger and vitamin B12 analog hydroxycobalamin (Cbl) as a new tool to investigate the involvement of endogenous H₂S in CB oxygen sensing. We observed that the slow-release sulfide donor GYY4137 elicited catecholamine release from isolated whole carotid bodies, and that Cbl prevented this response. Cbl also abolished the rise in [Ca2+]i evoked by 50 µM NaHS in enzymatically dispersed CB glomus cells. Moreover, Cbl markedly inhibited the catecholamine release and [Ca2+]i rise caused by hypoxia in isolated CBs and dispersed glomus cells, respectively, whereas it did not alter these responses when they were evoked by high [K⁺]e. The L-type Ca2+ channel blocker nifedipine slightly inhibited the rise in CB chemoreceptor cells [Ca2+]i elicited by sulfide, whilst causing a somewhat larger attenuation of the hypoxia-induced Ca2+ signal. We conclude that Cbl is a useful and specific tool for studying the function of H₂S in cells. Based on its effects on the CB chemoreceptor cells we propose that endogenous H₂S is an amplifier of the hypoxic transduction cascade which acts mainly by stimulating non-L-type Ca2+ channels.
Keywords: carotid body; hydrogen sulfide; hydroxycobalamin; hypoxia; oxygen sensing.
Conflict of interest statement
The authors declare no conflict of interest.
Figures

Similar articles
-
A crucial role for hydrogen sulfide in oxygen sensing via modulating large conductance calcium-activated potassium channels.Antioxid Redox Signal. 2010 May 15;12(10):1179-89. doi: 10.1089/ars.2009.2926. Antioxid Redox Signal. 2010. PMID: 19803741
-
Hydrogen sulfide and hypoxia-induced changes in TASK (K2P3/9) activity and intracellular Ca(2+) concentration in rat carotid body glomus cells.Respir Physiol Neurobiol. 2015 Aug 15;215:30-8. doi: 10.1016/j.resp.2015.04.012. Epub 2015 May 5. Respir Physiol Neurobiol. 2015. PMID: 25956223 Free PMC article.
-
Effects of low glucose on carotid body chemoreceptor cell activity studied in cultures of intact organs and in dissociated cells.Am J Physiol Cell Physiol. 2012 Apr 15;302(8):C1128-40. doi: 10.1152/ajpcell.00196.2011. Epub 2011 Dec 21. Am J Physiol Cell Physiol. 2012. PMID: 22189552
-
Carotid body chemoreception: the importance of CO2-HCO3- and carbonic anhydrase. (review).Biol Res. 1993;26(3):319-29. Biol Res. 1993. PMID: 7606251 Review.
-
Carotid body oxygen sensing and adaptation to hypoxia.Pflugers Arch. 2016 Jan;468(1):59-70. doi: 10.1007/s00424-015-1734-0. Epub 2015 Sep 16. Pflugers Arch. 2016. PMID: 26373853 Review.
Cited by
-
The Role of Hydrogen Sulfide in the Regulation of the Pulmonary Vasculature in Health and Disease.Antioxidants (Basel). 2025 Mar 14;14(3):341. doi: 10.3390/antiox14030341. Antioxidants (Basel). 2025. PMID: 40227402 Free PMC article. Review.
-
Recent Development of the Molecular and Cellular Mechanisms of Hydrogen Sulfide Gasotransmitter.Antioxidants (Basel). 2022 Sep 10;11(9):1788. doi: 10.3390/antiox11091788. Antioxidants (Basel). 2022. PMID: 36139861 Free PMC article. Review.
-
The cystathionine-γ-lyase inhibitor DL-propargylglycine augments the ability of L-cysteine ethyl ester to overcome the adverse effects of morphine on breathing.Am J Physiol Lung Cell Mol Physiol. 2025 Jun 1;328(6):L809-L825. doi: 10.1152/ajplung.00003.2025. Epub 2025 Mar 18. Am J Physiol Lung Cell Mol Physiol. 2025. PMID: 40099842 Free PMC article.
-
Methods for Suppressing Hydrogen Sulfide in Biological Systems.Antioxid Redox Signal. 2022 Feb;36(4-6):294-308. doi: 10.1089/ars.2021.0088. Epub 2021 Aug 2. Antioxid Redox Signal. 2022. PMID: 34162216 Free PMC article. Review.
-
Mitochondrial Succinate Metabolism and Reactive Oxygen Species Are Important but Not Essential for Eliciting Carotid Body and Ventilatory Responses to Hypoxia in the Rat.Antioxidants (Basel). 2021 May 25;10(6):840. doi: 10.3390/antiox10060840. Antioxidants (Basel). 2021. PMID: 34070267 Free PMC article.
References
-
- Fidone S., Gonzalez C. Initiation and control of chemoreceptor activity in the carotid body. In: Fishman A.P., Cherniack N.S., Widdicombe J.G., Geiger S.R., editors. Handbook of Physiology, Section 3, The Respiratory System, vol II, Control of Breathing. Volume 2. American Physiological Society; Bethesda, MD, USA: 1986. pp. 247–312.
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

