NLRP3 Inflammasome and Inflammatory Bowel Disease
- PMID: 30873162
- PMCID: PMC6403142
- DOI: 10.3389/fimmu.2019.00276
NLRP3 Inflammasome and Inflammatory Bowel Disease
Abstract
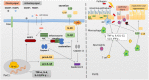
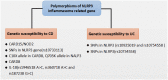
NLRP3 inflammasome can be widely found in epithelial cells and immune cells. The NOD-like receptors (NLRs) family member NLRP3 contains a central nucleotide-binding and oligomerization (NACHT) domain which facilitates self-oligomerization and has ATPase activity. The C-terminal conserves a leucine-rich repeats (LRRs) domain which can modulate NLRP3 activity and sense endogenous alarmins and microbial ligands. In contrast, the N-terminal pyrin domain (PYD) can account for homotypic interactions with the adaptor protein-ASC of NLRP3 inflammasome. These characters enable it function in innate immunity. Its downstream effector proteins include caspase-1 and IL-1β etc. which exhibit protective or detrimental roles in mucosal immunity in different studies. Here, we comprehensively review the current literature regarding the physiology of NLRP3 inflammasome and its potential roles in the pathogenesis of IBD. We also discuss about the complex interactions among the NLRP3 inflammasome, mucosal immune response, and gut homeostasis as found in experimental models and IBD patients.
Keywords: Crohn's disease; NLRP3 inflammasome; gut homeostasis; mucosal immune; ulcerative colitis.
Figures
References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

