β3 -Adrenoceptor as a potential immuno-suppressor agent in melanoma
- PMID: 30874296
- PMCID: PMC6592854
- DOI: 10.1111/bph.14660
β3 -Adrenoceptor as a potential immuno-suppressor agent in melanoma
Abstract
Background and purpose: Stress-related catecholamines have a role in cancer and β-adrenoceptors; specifically, β2 -adrenoceptors have been identified as new targets in treating melanoma. Recently, β3 -adrenoceptors have shown a pleiotropic effect on melanoma micro-environment leading to cancer progression. However, the mechanisms by which β3 -adrenoceptors promote this progression remain poorly understood. Catecholamines affect the immune system by modulating several factors that can alter immune cell sub-population homeostasis. Understanding the mechanisms of cancer immune-tolerance is one of the most intriguing challenges in modern research. This study investigates the potential role of β3 -adrenoceptors in immune-tolerance regulation.
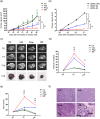
Experimental approach: A mouse model of melanoma in which syngeneic B16-F10 cells were injected in C57BL-6 mice was used to evaluate the effect of β-adrenoceptor blockade on the number and activity of immune cell sub-populations (Treg, NK, CD8, MDSC, macrophages, and neutrophils). Pharmacological and molecular approaches with β-blockers (propranolol and SR59230A) and specific β-adrenoceptor siRNAs targeting β2 - or β3 -adrenoceptors were used.
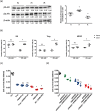
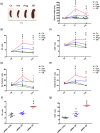
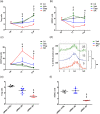
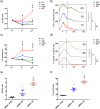
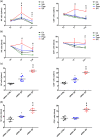
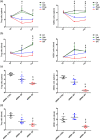
Key results: Only β3 -, but not β2 -adrenoceptors, were up-regulated under hypoxia in peripheral blood mononuclear cells and selectively expressed in immune cell sub-populations including Treg, MDSC, and NK. SR59230A and β3 -adrenoceptor siRNAs increased NK and CD8 number and cytotoxicity, while they attenuated Treg and MDSC sub-populations in the tumour mass, blood, and spleen. SR59230A and β3 -adrenoceptor siRNAs increased the ratio of M1/M2 macrophages and N1 granulocytes.
Conclusions and implications: Our data suggest that β3 -adrenoceptors are involved in immune-tolerance, which opens the way for new strategic therapies to overcome melanoma growth.
Linked articles: This article is part of a themed section on Adrenoceptors-New Roles for Old Players. To view the other articles in this section visit http://onlinelibrary.wiley.com/doi/10.1111/bph.v176.14/issuetoc.
© 2019 The British Pharmacological Society.
Conflict of interest statement
The authors declare no conflicts of interest.
Figures
Similar articles
-
β-Adrenoceptors as drug targets in melanoma: novel preclinical evidence for a role of β3 -adrenoceptors.Br J Pharmacol. 2019 Jul;176(14):2496-2508. doi: 10.1111/bph.14552. Epub 2018 Dec 18. Br J Pharmacol. 2019. PMID: 30471093 Free PMC article. Review.
-
Stimulation of β-adrenoceptors up-regulates cardiac expression of galectin-3 and BIM through the Hippo signalling pathway.Br J Pharmacol. 2019 Jul;176(14):2465-2481. doi: 10.1111/bph.14674. Epub 2019 May 30. Br J Pharmacol. 2019. PMID: 30932177 Free PMC article.
-
Agonist-induced desensitisation of β3 -adrenoceptors: Where, when, and how?Br J Pharmacol. 2019 Jul;176(14):2539-2558. doi: 10.1111/bph.14633. Epub 2019 Apr 7. Br J Pharmacol. 2019. PMID: 30809805 Free PMC article. Review.
-
Functional involvement of β3-adrenergic receptors in melanoma growth and vascularization.J Mol Med (Berl). 2013 Dec;91(12):1407-19. doi: 10.1007/s00109-013-1073-6. Epub 2013 Aug 2. J Mol Med (Berl). 2013. PMID: 23907236
-
Beta Blockers and Melanoma.Open Access Maced J Med Sci. 2019 Aug 30;7(18):3110-3112. doi: 10.3889/oamjms.2019.781. eCollection 2019 Sep 30. Open Access Maced J Med Sci. 2019. PMID: 31850134 Free PMC article. Review.
Cited by
-
Neuroendocrine Factors in Melanoma Pathogenesis.Cancers (Basel). 2021 May 10;13(9):2277. doi: 10.3390/cancers13092277. Cancers (Basel). 2021. PMID: 34068618 Free PMC article. Review.
-
Decoupling Oxygen Tension From Retinal Vascularization as a New Perspective for Management of Retinopathy of Prematurity. New Opportunities From β-adrenoceptors.Front Pharmacol. 2022 Jan 21;13:835771. doi: 10.3389/fphar.2022.835771. eCollection 2022. Front Pharmacol. 2022. PMID: 35126166 Free PMC article. Review.
-
Hypoxia and Extracellular Acidification as Drivers of Melanoma Progression and Drug Resistance.Cells. 2021 Apr 9;10(4):862. doi: 10.3390/cells10040862. Cells. 2021. PMID: 33918883 Free PMC article. Review.
-
The Genetic Basis of Dormancy and Awakening in Cutaneous Metastatic Melanoma.Cancers (Basel). 2022 Apr 23;14(9):2104. doi: 10.3390/cancers14092104. Cancers (Basel). 2022. PMID: 35565234 Free PMC article. Review.
-
Everything You Always Wanted to Know about β3-AR * (* But Were Afraid to Ask).Cells. 2019 Apr 16;8(4):357. doi: 10.3390/cells8040357. Cells. 2019. PMID: 30995798 Free PMC article. Review.
References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Research Materials
Miscellaneous

