The Role of Primary Cilia in the Crosstalk between the Ubiquitin⁻Proteasome System and Autophagy
- PMID: 30875746
- PMCID: PMC6468794
- DOI: 10.3390/cells8030241
The Role of Primary Cilia in the Crosstalk between the Ubiquitin⁻Proteasome System and Autophagy
Abstract
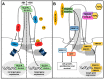
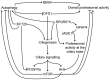
Protein degradation is a pivotal process for eukaryotic development and homeostasis. The majority of proteins are degraded by the ubiquitin⁻proteasome system and by autophagy. Recent studies describe a crosstalk between these two main eukaryotic degradation systems which allows for establishing a kind of safety mechanism. If one of these degradation systems is hampered, the other compensates for this defect. The mechanism behind this crosstalk is poorly understood. Novel studies suggest that primary cilia, little cellular protrusions, are involved in the regulation of the crosstalk between the two degradation systems. In this review article, we summarise the current knowledge about the association between cilia, the ubiquitin⁻proteasome system and autophagy.
Keywords: BBS4; GLI; IFT; OFD1; RPGRIP1L; hedgehog; mTOR; neurodegenerative diseases; protein aggregation.
Conflict of interest statement
The authors declare that they have no competing interests.
Figures

References
-
- Cox D., Raeburn C., Sui X., Hatters D. Seminars in Cell & Developmental Biology. Academic Press; New York, NY, USA: 2018. Protein aggregation in cell biology: An aggregomics perspective of health and disease. pii: S1084-9521. - PubMed
-
- Koepp D. Cell cycle regulation by protein degradation. Methods Mol. Biol. 2014;1170:61–73. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Miscellaneous

