Clinical follow-up on weight loss, glycemic control, and safety aspects of 24 months of duodenal-jejunal bypass liner implantation
- PMID: 30877567
- PMCID: PMC6946747
- DOI: 10.1007/s00464-019-06752-8
Clinical follow-up on weight loss, glycemic control, and safety aspects of 24 months of duodenal-jejunal bypass liner implantation
Abstract
Background: The duodenal-jejunal bypass liner (DJBL) is an endoscopic device designed to induce weight loss and improve glycemic control. The liner is licensed for a maximum implant duration of 12 months. It might be hypothesized that extension of the dwelling time results in added value. The goals of our study were to determine weight change, change in glycemic control, and safety in patients with an intended 24 months of DJBL dwelling time.
Methods: Patients were initially selected for a 12-month implantation period. When no physical complaints or adverse events (AEs) occurred, motivated patients who responded well were selected for extension of dwelling time to 24 months. Patients underwent a control endoscopy 12 months after implantation and visited the out-patient clinic every 3 months up to explantation. Patients agreed to remove the DJBL when complaints or AEs occurred that could not be treated conservatively.
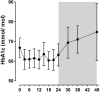
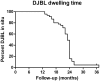
Results: Implantation was extended in 44 patients, and 24 (55%) patients completed the full 24 months. Twenty patients required early removal due to AEs. During dwelling time, body weight decreased significantly (15.9 kg; TBWL 14.6%). HbA1c decreased non-significantly (4.9 mmol/mol). The number of insulin users and daily dose of insulin both decreased significantly. At 24 months after removal, glycemic control had worsened, while body weight was still significantly lower compared to baseline. In total, 68% of the patients experienced at least one AE. Two patients developed a hepatic abscess.
Conclusions: DJBL treatment results in significant weight loss and improves glycemic control during implantation. The largest beneficial effects occur during the first 9-12 months after implantation. Extension of dwelling time to 24 months results only in stabilization of body weight and glycemic control. After explantation, weight improvements are maintained, but glycemic control worsens. As the cumulative risk of AEs increases with time, a maximal dwelling time of 12 months is advisable.
Keywords: Adverse events; Diabetes mellitus; Duodenal-jejunal bypass liner; Endobarrier; Hepatic abscess; Migration.
Conflict of interest statement
Dr. E.O. Aarts, and dr. P.J. Wahab received support for travel as proctor from GI Dynamics. Drs. Janssen received financial research support and consultancy fees from GI Dynamics. Drs. B. Betzel, drs. M. Cooiman, dr. M.J.M. Groenen, dr. J.P.H. Drenth, and dr. F.J. Berends have no conflicts of interests of financial ties to disclose.
Figures


References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Research Materials

