Variations in the secondary structures of PAM proteins influence their binding affinities to human plasminogen
- PMID: 30880082
- PMCID: PMC6502667
- DOI: 10.1016/j.jsb.2019.03.003
Variations in the secondary structures of PAM proteins influence their binding affinities to human plasminogen
Abstract
M-proteins (M-Prts) are major virulence determinants of Group A Streptococcus pyogenes (GAS) that are covalently anchored to the cell wall at their conserved COOH-termini while the NH2-terminal regions extend through the capsule into extracellular space. Functional M-Prts are also secreted and/or released from GAS cells where they exist as helical coiled-coil dimers in solution. Certain GAS strains (Pattern D) uniquely express an M-protein (plasminogen-binding group A streptococcal M-protein; PAM) that directly interacts with human plasminogen (hPg), a process strongly implicated in the virulence of these strains. M-Prt expressed by the emm gene is employed to serotype over 250 known strains of GAS, ∼20 of which are hitherto found to express PAMs. We have developed a modular structural model of the PAM dimer that describes the roles of different domains of this protein in various functions. While the helical COOH-terminal domains of PAM are essential for dimerization in solution, regions of its NH2-terminal domains also exhibit a weak potential to dimerize. We find that temperature controls the open (unwound) or closed (wound) states of the functional NH2-terminal domains of PAM. As temperature increases, α-helices are dramatically reduced, which concomitantly destabilizes the helical coiled-coil PAM dimers. PAMs with two a-repeats within the variable NH2-terminal A-domain (class I/III) bind to hPg tightly, but natural PAM isolates with a single a-repeat in this domain (class II) display dramatic changes in hPg binding with temperature. We conclude that coexistence of two a-repeats in PAM is critical to achieve optimal binding to hPg, especially in its monomeric form, at the biologically relevant temperature.
Keywords: Coiled coils; Dimer dissociation; PAM dimerization; Plasminogen acquisition; Single a-repeat; α-Helix.
Copyright © 2019 Elsevier Inc. All rights reserved.
Conflict of interest statement
Conflicts of Interest
The authors declare that they have no conflicts of interest.
Figures
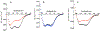




Similar articles
-
A local α-helix drives structural evolution of streptococcal M-protein affinity for host human plasminogen.Biochem J. 2020 May 15;477(9):1613-1630. doi: 10.1042/BCJ20200197. Biochem J. 2020. PMID: 32270857 Free PMC article.
-
Contributions of different modules of the plasminogen-binding Streptococcus pyogenes M-protein that mediate its functional dimerization.J Struct Biol. 2018 Nov;204(2):151-164. doi: 10.1016/j.jsb.2018.07.017. Epub 2018 Jul 30. J Struct Biol. 2018. PMID: 30071314 Free PMC article.
-
Dimerization is not a determining factor for functional high affinity human plasminogen binding by the group A streptococcal virulence factor PAM and is mediated by specific residues within the PAM a1a2 domain.J Biol Chem. 2014 Aug 1;289(31):21684-93. doi: 10.1074/jbc.M114.570218. Epub 2014 Jun 24. J Biol Chem. 2014. PMID: 24962580 Free PMC article.
-
Binding of the kringle-2 domain of human plasminogen to streptococcal PAM-type M-protein causes dissociation of PAM dimers.Microbiologyopen. 2021 Nov;10(6):e1252. doi: 10.1002/mbo3.1252. Microbiologyopen. 2021. PMID: 34964287 Free PMC article.
-
Pathogen activators of plasminogen.J Thromb Haemost. 2015 Jun;13 Suppl 1(0 1):S106-14. doi: 10.1111/jth.12939. J Thromb Haemost. 2015. PMID: 26149011 Free PMC article. Review.
Cited by
-
Inactivation of the lysine binding sites of human plasminogen (hPg) reveals novel structural requirements for the tight hPg conformation, M-protein binding, and rapid activation.Front Mol Biosci. 2023 Apr 4;10:1166155. doi: 10.3389/fmolb.2023.1166155. eCollection 2023. Front Mol Biosci. 2023. PMID: 37081852 Free PMC article.
-
The M Protein of Streptococcus pyogenes Strain AP53 Retains Cell Surface Functional Plasminogen Binding after Inactivation of the Sortase A Gene.J Bacteriol. 2020 Apr 27;202(10):e00096-20. doi: 10.1128/JB.00096-20. Print 2020 Apr 27. J Bacteriol. 2020. PMID: 32123038 Free PMC article.
-
A local α-helix drives structural evolution of streptococcal M-protein affinity for host human plasminogen.Biochem J. 2020 May 15;477(9):1613-1630. doi: 10.1042/BCJ20200197. Biochem J. 2020. PMID: 32270857 Free PMC article.
-
Streptococcus co-opts a conformational lock in human plasminogen to facilitate streptokinase cleavage and bacterial virulence.J Biol Chem. 2021 Jan-Jun;296:100099. doi: 10.1074/jbc.RA120.016262. Epub 2020 Nov 24. J Biol Chem. 2021. PMID: 33208461 Free PMC article.
-
Relationships Between Plasminogen-Binding M-Protein and Surface Enolase for Human Plasminogen Acquisition and Activation in Streptococcus pyogenes.Front Microbiol. 2022 May 24;13:905670. doi: 10.3389/fmicb.2022.905670. eCollection 2022. Front Microbiol. 2022. PMID: 35685926 Free PMC article.
References
-
- Akerström B, Lindah G, Björck L, Lindqvist A, 1992. Protein Arp and protein H from group A streptococci. Ig binding and dimerization are regulated by temperature. J. Immunol 148, 3238–3243. - PubMed
-
- Babul J, Stellwagen E, 1969. Measurement of protein concentration with interference optics. Anal. Biochem 28, 216–221. - PubMed
-
- Berge A, Sjobring U, 1993. PAM, a novel plasminogen-binding protein from Streptococcus pyogenes. J. Biol. Chem 268, 25417–25424. - PubMed
-
- Berge A, Björck L, 1995. Streptococcal cysteine proteinase releases biologically active fragments of streptococcal surface proteins. J. Biol. Chem 270, 9862–9867. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

