Methoxy substituted 2-benzylidene-1-indanone derivatives as A1 and/or A2A AR antagonists for the potential treatment of neurological conditions
- PMID: 30881617
- PMCID: PMC6390816
- DOI: 10.1039/c8md00540k
Methoxy substituted 2-benzylidene-1-indanone derivatives as A1 and/or A2A AR antagonists for the potential treatment of neurological conditions
Abstract
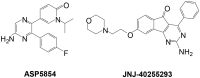
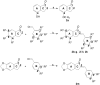
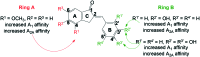
A prior study reported on hydroxy substituted 2-benzylidene-1-indanone derivatives as A1 and/or A2A antagonists for the potential treatment of neurological conditions. A lead compound (1a) was identified with both A1 and A2A affinity in the micromolar range. The current study explored the structurally related methoxy substituted 2-benzylidene-1-indanone derivatives with various substitutions on ring A and B of the benzylidene indanone scaffold in order to enhance A1 and A2A affinity. This led to compounds with both A1 and A2A affinity in the nanomolar range, namely 2c (A1 K i (rat) = 41 nM; A2A K i (rat) = 97 nM) with C4-OCH3 substitution on ring A together with meta (3') hydroxy substitution on ring B and 2e (A1 K i (rat) = 42 nM; A2A K i (rat) = 78 nM) with C4-OCH3 substitution on ring A together with meta (3') and para (4') dihydroxy substitution on ring B. Additionally, 2c is an A1 antagonist. Consequently, the methoxy substituted 2-benzylidene-1-indanone scaffold is highly promising for the design of novel A1 and A2A antagonists.
Figures

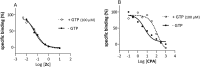
Similar articles
-
2-Benzylidene-1-Indanone Analogues as Dual Adenosine A1/A2a Receptor Antagonists for the Potential Treatment of Neurological Conditions.Drug Res (Stuttg). 2019 Jul;69(7):382-391. doi: 10.1055/a-0808-3993. Epub 2019 Jan 7. Drug Res (Stuttg). 2019. PMID: 30616250
-
Synthesis and Structure Activity Relationships of Chalcone based Benzocycloalkanone Derivatives as Adenosine A1 and/or A2A Receptor Antagonists.Drug Res (Stuttg). 2020 Jun;70(6):243-256. doi: 10.1055/a-1146-2996. Epub 2020 Apr 29. Drug Res (Stuttg). 2020. PMID: 32349128
-
5-Substituted 2-benzylidene-1-tetralone analogues as A1 and/or A2A antagonists for the potential treatment of neurological conditions.Bioorg Chem. 2017 Oct;74:251-259. doi: 10.1016/j.bioorg.2017.08.013. Epub 2017 Aug 30. Bioorg Chem. 2017. PMID: 28881253
-
Evaluation of 2-benzylidene-1-tetralone derivatives as antagonists of A1 and A2A adenosine receptors.Chem Biol Drug Des. 2018 Jan;91(1):234-244. doi: 10.1111/cbdd.13074. Epub 2017 Aug 16. Chem Biol Drug Des. 2018. PMID: 28734058
-
Selective adenosine A2A receptor antagonists.Farmaco. 2001 Jan-Feb;56(1-2):87-90. doi: 10.1016/s0014-827x(01)01024-2. Farmaco. 2001. PMID: 11347973 Review.
Cited by
-
Dual A1 and A2A adenosine receptor antagonists, methoxy substituted 2-benzylidene-1-indanone, suppresses intestinal postprandial glucose and attenuates hyperglycaemia in fructose-streptozotocin diabetic rats.BMC Endocr Disord. 2023 May 4;23(1):97. doi: 10.1186/s12902-023-01354-x. BMC Endocr Disord. 2023. PMID: 37143025 Free PMC article.
-
C3 amino-substituted chalcone derivative with selective adenosine rA1 receptor affinity in the micromolar range.Chem Zvesti. 2021;75(4):1581-1605. doi: 10.1007/s11696-020-01414-9. Epub 2020 Nov 17. Chem Zvesti. 2021. PMID: 33223599 Free PMC article.
-
Chalcones as Potential Ligands for the Treatment of Parkinson's Disease.Pharmaceuticals (Basel). 2022 Jul 10;15(7):847. doi: 10.3390/ph15070847. Pharmaceuticals (Basel). 2022. PMID: 35890146 Free PMC article. Review.
-
Untargeted Metabolomics Reveals the Potential Antidepressant Activity of a Novel Adenosine Receptor Antagonist.Molecules. 2022 Mar 24;27(7):2094. doi: 10.3390/molecules27072094. Molecules. 2022. PMID: 35408500 Free PMC article.
-
Exploration of chalcones and related heterocycle compounds as ligands of adenosine receptors: therapeutics development.Mol Divers. 2022 Jun;26(3):1779-1821. doi: 10.1007/s11030-021-10257-9. Epub 2021 Jun 27. Mol Divers. 2022. PMID: 34176057 Review.
References
-
- Soliman A. M., Fathalla A. M., Moustafa A. A. Pharmacol. Rep. 2018;70(4):661–667. - PubMed
-
- Palmer T. M., Stiles G. L. Neuropharmacology. 1995;34(7):683–694. - PubMed
-
- Ross G. W., Abbot R. D., Petrovitch H., Morens D. M., Grandinetti A., Tung K. H., Tanner C. M., Masaki K. H., Blanchette P. L., Curb J. D., Popper J. S. JAMA, J. Am. Med. Assoc. 2000;283(20):2674–2679. - PubMed
-
- Prediger R. D. S., Ribeiro J. A., Sebastião A. M. J. Alzheimers Dis. J. Alzheimer's Dis. 2010;2010;2020(S1)(S1):S205–S2205. S3–S15. - PubMed
-
- Ribeiro J. A., Sebastião A. M. J. Alzheimer's Dis. 2010;20(S1):S3–S15. - PubMed
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

