Novel endosomal NOX2 oxidase inhibitor ameliorates pandemic influenza A virus-induced lung inflammation in mice
- PMID: 30884042
- PMCID: PMC6972593
- DOI: 10.1111/resp.13524
Novel endosomal NOX2 oxidase inhibitor ameliorates pandemic influenza A virus-induced lung inflammation in mice
Abstract
Background and objective: Influenza A viruses (IAV) cause respiratory tract infections that can be fatal when the virus spreads to the alveolar space (i.e. alveolitis), and this is mainly observed with highly pathogenic strains. Reactive oxygen species (ROS) production by the NOX2 NADPH oxidase in endosomes has been directly implicated in IAV pathology. Recently, we demonstrated that treatment with a novel endosome-targeted NOX2 oxidase inhibitor, cholestanol-conjugated gp91dsTAT (Cgp91ds-TAT), attenuated airway inflammation and viral replication to infection with a low pathogenic influenza A viral strain. Here, we determined whether suppression of endosome NOX2 oxidase prevents the lung inflammation following infection with a highly pathogenic IAV strain.
Methods: C57Bl/6 mice were intranasally treated with either DMSO vehicle (2%) or Cgp91ds-TAT (0.2 mg/kg/day) 1 day prior to infection with the high pathogenicity PR8 IAV strain (500 PFU/mouse). At Day 3 post-infection, mice were culled for the evaluation of airway and lung inflammation, viral titres and ROS generation.
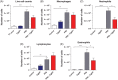
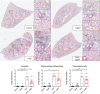
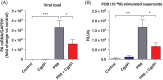
Results: PR8 infection resulted in a marked degree of airway inflammation, epithelial denudation, alveolitis and inflammatory cell ROS production. Cgp91ds-TAT treatment significantly attenuated airway inflammation, including neutrophil influx, the degree of alveolitis and inflammatory cell ROS generation. Importantly, the anti-inflammatory phenotype affected by Cgp91ds-TAT significantly enhanced the clearance of lung viral mRNA following PR8 infection.
Conclusion: Endosomal NOX2 oxidase promotes pathogenic lung inflammation to IAV infection. The localized delivery of endosomal NOX2 oxidase inhibitors is a novel therapeutic strategy against IAV, which has the potential to limit the pathogenesis caused during epidemics and pandemics.
Keywords: NOX2 oxidase; endosome; inflammation; influenza; respiratory infections.
© 2019 Asian Pacific Society of Respirology.
Figures
Comment in
-
Targeting endosomal NOX2 as a potential therapy to limit influenza A virus infection.Respirology. 2019 Oct;24(10):935-936. doi: 10.1111/resp.13629. Epub 2019 Jun 21. Respirology. 2019. PMID: 31225681 No abstract available.
Similar articles
-
Inhibition of Nox2 oxidase activity ameliorates influenza A virus-induced lung inflammation.PLoS Pathog. 2011 Feb 3;7(2):e1001271. doi: 10.1371/journal.ppat.1001271. PLoS Pathog. 2011. PMID: 21304882 Free PMC article.
-
Therapeutic Targeting of Endosome and Mitochondrial Reactive Oxygen Species Protects Mice From Influenza Virus Morbidity.Front Pharmacol. 2022 Mar 23;13:870156. doi: 10.3389/fphar.2022.870156. eCollection 2022. Front Pharmacol. 2022. PMID: 35401240 Free PMC article.
-
Endosomal NOX2 oxidase exacerbates virus pathogenicity and is a target for antiviral therapy.Nat Commun. 2017 Jul 12;8(1):69. doi: 10.1038/s41467-017-00057-x. Nat Commun. 2017. PMID: 28701733 Free PMC article.
-
NADPH oxidases as novel pharmacologic targets against influenza A virus infection.Mol Pharmacol. 2014 Dec;86(6):747-59. doi: 10.1124/mol.114.095216. Epub 2014 Oct 9. Mol Pharmacol. 2014. PMID: 25301784 Review.
-
L-carnosine modulates respiratory burst and reactive oxygen species production in neutrophil biochemistry and function: may oral dosage form of non-hydrolized dipeptide L-carnosine complement anti-infective anti-influenza flu treatment, prevention and self-care as an alternative to the conventional vaccination?Curr Clin Pharmacol. 2014 May;9(2):93-115. doi: 10.2174/1574884709999140311125601. Curr Clin Pharmacol. 2014. PMID: 23441838 Review.
Cited by
-
Severity of neonatal influenza infection is driven by type I interferon and oxidative stress.Mucosal Immunol. 2022 Jun;15(6):1309-1320. doi: 10.1038/s41385-022-00576-x. Epub 2022 Nov 9. Mucosal Immunol. 2022. PMID: 36352099 Free PMC article.
-
EROS is a selective chaperone regulating the phagocyte NADPH oxidase and purinergic signalling.Elife. 2022 Nov 24;11:e76387. doi: 10.7554/eLife.76387. Elife. 2022. PMID: 36421765 Free PMC article.
-
Global m6A methylation and gene expression patterns in human microglial HMC3 cells infected with HIV-1.Heliyon. 2023 Oct 26;9(11):e21307. doi: 10.1016/j.heliyon.2023.e21307. eCollection 2023 Nov. Heliyon. 2023. PMID: 38027859 Free PMC article.
-
COVID-19: Proposing a Ketone-Based Metabolic Therapy as a Treatment to Blunt the Cytokine Storm.Oxid Med Cell Longev. 2020 Sep 9;2020:6401341. doi: 10.1155/2020/6401341. eCollection 2020. Oxid Med Cell Longev. 2020. PMID: 33014275 Free PMC article. Review.
-
Redox control in the pathophysiology of influenza virus infection.BMC Microbiol. 2020 Jul 20;20(1):214. doi: 10.1186/s12866-020-01890-9. BMC Microbiol. 2020. PMID: 32689931 Free PMC article. Review.
References
-
- Moscona A. Neuraminidase inhibitors for influenza. N. Engl. J. Med. 2005; 353: 1363–73. - PubMed
-
- Nitsch‐Osuch A, Brydak LB. Treatment and prophylaxis of influenza and the problem of resistance to neuraminidase inhibitors. Postepy Hig. Med. Dosw. (Online) 2015; 69: 1087–95. - PubMed
-
- Schwarz KB. Oxidative stress during viral infection: a review. Free Radic. Biol. Med. 1996; 21: 641–9. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

