Loss of the TAM Receptor Axl Ameliorates Severe Zika Virus Pathogenesis and Reduces Apoptosis in Microglia
- PMID: 30884311
- PMCID: PMC6424058
- DOI: 10.1016/j.isci.2019.03.003
Loss of the TAM Receptor Axl Ameliorates Severe Zika Virus Pathogenesis and Reduces Apoptosis in Microglia
Abstract
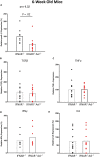
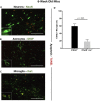
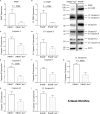
The TAM receptor, Axl, has been implicated as a candidate entry receptor for Zika virus (ZIKV) infection but has been shown as inessential for virus infection in mice. To probe the role of Axl in murine ZIKV infection, we developed a mouse model lacking the Axl receptor and the interferon alpha/beta receptor (Ifnar-/-Axl-/-), conferring susceptibility to ZIKV. This model validated that Axl is not required for murine ZIKV infection and that mice lacking Axl are resistant to ZIKV pathogenesis. This resistance correlates to lower pro-interleukin-1β production and less apoptosis in microglia of ZIKV-infected mice. This apoptosis occurs through both intrinsic (caspase 9) and extrinsic (caspase 8) manners, and is age dependent, as younger Axl-deficient mice are susceptible to ZIKV pathogenesis. These findings suggest that Axl plays an important role in pathogenesis in the brain during ZIKV infection and indicates a potential role for Axl inhibitors as therapeutics during viral infection.
Keywords: Biological Sciences; Neurotoxicology; Virology.
Copyright © 2019 The Author(s). Published by Elsevier Inc. All rights reserved.
Figures



Similar articles
-
TAM Receptors Are Not Required for Zika Virus Infection in Mice.Cell Rep. 2017 Apr 18;19(3):558-568. doi: 10.1016/j.celrep.2017.03.058. Cell Rep. 2017. PMID: 28423319 Free PMC article.
-
AXL promotes Zika virus infection in astrocytes by antagonizing type I interferon signalling.Nat Microbiol. 2018 Mar;3(3):302-309. doi: 10.1038/s41564-017-0092-4. Epub 2018 Jan 29. Nat Microbiol. 2018. PMID: 29379210
-
AXL receptor is required for Zika virus strain MR-766 infection in human glioblastoma cell lines.Mol Ther Oncolytics. 2021 Nov 11;23:447-457. doi: 10.1016/j.omto.2021.11.001. eCollection 2021 Dec 17. Mol Ther Oncolytics. 2021. PMID: 34901388 Free PMC article.
-
Genetic Diversity of Collaborative Cross Mice Controls Viral Replication, Clinical Severity, and Brain Pathology Induced by Zika Virus Infection, Independently of Oas1b.J Virol. 2020 Jan 17;94(3):e01034-19. doi: 10.1128/JVI.01034-19. Print 2020 Jan 17. J Virol. 2020. PMID: 31694939 Free PMC article.
-
Infection by Zika viruses requires the transmembrane protein AXL, endocytosis and low pH.Virology. 2018 May;518:301-312. doi: 10.1016/j.virol.2018.03.009. Epub 2018 Mar 22. Virology. 2018. PMID: 29574335 Free PMC article.
Cited by
-
Axl Deficiency Promotes the Neuroinvasion of Japanese Encephalitis Virus by Enhancing IL-1α Production from Pyroptotic Macrophages.J Virol. 2020 Aug 17;94(17):e00602-20. doi: 10.1128/JVI.00602-20. Print 2020 Aug 17. J Virol. 2020. PMID: 32611752 Free PMC article.
-
Monodelphis domestica as a Fetal Intra-Cerebral Inoculation Model for Zika Virus Pathogenesis.Pathogens. 2023 May 19;12(5):733. doi: 10.3390/pathogens12050733. Pathogens. 2023. PMID: 37242404 Free PMC article.
-
Crosstalk between RNA Metabolism and Cellular Stress Responses during Zika Virus Replication.Pathogens. 2020 Feb 25;9(3):158. doi: 10.3390/pathogens9030158. Pathogens. 2020. PMID: 32106582 Free PMC article. Review.
-
Japanese Encephalitis Virus NS2B-3 Protein Complex Promotes Cell Apoptosis and Viral Particle Release by Down-Regulating the Expression of AXL.Virol Sin. 2021 Dec;36(6):1503-1519. doi: 10.1007/s12250-021-00442-3. Epub 2021 Sep 6. Virol Sin. 2021. PMID: 34487337 Free PMC article.
-
Molecular and Cellular Mechanisms Underlying Neurologic Manifestations of Mosquito-Borne Flavivirus Infections.Viruses. 2023 Oct 31;15(11):2200. doi: 10.3390/v15112200. Viruses. 2023. PMID: 38005878 Free PMC article. Review.
References
-
- Bearcroft W.G. Zika virus infection experimentally induced in a human volunteer. Trans. R. Soc. Trop. Med. Hyg. 1956;50:442–448. - PubMed
-
- Brown G.C., Neher J.J. Microglial phagocytosis of live neurons. Nat. Rev. Neurosci. 2014;15:209. - PubMed
-
- Caraux A., Kim N., Bell S.E., Zompi S., Ranson T., Lesjean-Pottier S., Garcia-Ojeda M.E., Turner M., Colucci F. Phospholipase C-gamma2 is essential for NK cell cytotoxicity and innate immunity to malignant and virally infected cells. Blood. 2006;107:994–1002. - PubMed
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials
Miscellaneous

