New insights into the metal-induced oxidative degradation pathways of transthyretin
- PMID: 30887985
- PMCID: PMC6452628
- DOI: 10.1039/c9cc00682f
New insights into the metal-induced oxidative degradation pathways of transthyretin
Abstract
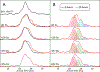
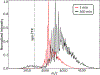
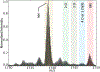
The amyloidogenic mechanism of transthyretin is still debated but understanding it fully could lend insight into disease progression and potential therapeutics. Transthyretin was investigated revealing a metal-induced (Cr/Cu) oxidation pathway leading to N-terminal backbone fragmentation and oligomer formation; previously hidden details were revealed only by FT-IM-Orbitrap MS and surface-induced dissociation.
Conflict of interest statement
Conflicts of interest
There are no conflicts to declare.
Figures


Similar articles
-
Effect of nitric oxide in amyloid fibril formation on transthyretin-related amyloidosis.Biochemistry. 2005 Aug 23;44(33):11122-9. doi: 10.1021/bi050327i. Biochemistry. 2005. PMID: 16101296
-
Disruption of the CD Loop by Enzymatic Cleavage Promotes the Formation of Toxic Transthyretin Oligomers through a Common Transthyretin Misfolding Pathway.Biochemistry. 2020 Jun 30;59(25):2319-2327. doi: 10.1021/acs.biochem.0c00079. Epub 2020 Jun 14. Biochemistry. 2020. PMID: 32500705 Free PMC article.
-
Topological Analysis of Transthyretin Disassembly Mechanism: Surface-Induced Dissociation Reveals Hidden Reaction Pathways.Anal Chem. 2019 Feb 5;91(3):2345-2351. doi: 10.1021/acs.analchem.8b05066. Epub 2019 Jan 28. Anal Chem. 2019. PMID: 30642177 Free PMC article.
-
Transthyretin Misfolding, A Fatal Structural Pathogenesis Mechanism.Int J Mol Sci. 2021 Apr 23;22(9):4429. doi: 10.3390/ijms22094429. Int J Mol Sci. 2021. PMID: 33922648 Free PMC article. Review.
-
Transthyretin quaternary and tertiary structural changes facilitate misassembly into amyloid.Adv Protein Chem. 1997;50:161-81. doi: 10.1016/s0065-3233(08)60321-6. Adv Protein Chem. 1997. PMID: 9338081 Review.
Cited by
-
Variable-Temperature Electrospray Ionization for Temperature-Dependent Folding/Refolding Reactions of Proteins and Ligand Binding.Anal Chem. 2021 May 11;93(18):6924-6931. doi: 10.1021/acs.analchem.1c00870. Epub 2021 Apr 27. Anal Chem. 2021. PMID: 33904705 Free PMC article.
-
Structural Analysis of the Effect of a Dual-FLAG Tag on Transthyretin.Biochemistry. 2020 Mar 10;59(9):1013-1022. doi: 10.1021/acs.biochem.0c00105. Epub 2020 Mar 2. Biochemistry. 2020. PMID: 32101399 Free PMC article.
-
Aggregation-Prone Structural Ensembles of Transthyretin Collected With Regression Analysis for NMR Chemical Shift.Front Mol Biosci. 2021 Oct 20;8:766830. doi: 10.3389/fmolb.2021.766830. eCollection 2021. Front Mol Biosci. 2021. PMID: 34746240 Free PMC article.
-
Native IM-Orbitrap MS: Resolving What Was Hidden.Trends Analyt Chem. 2020 Mar;124:115533. doi: 10.1016/j.trac.2019.05.035. Epub 2019 May 31. Trends Analyt Chem. 2020. PMID: 32189816 Free PMC article.
-
Implementing Digital-Waveform Technology for Extended m/z Range Operation on a Native Dual-Quadrupole FT-IM-Orbitrap Mass Spectrometer.J Am Soc Mass Spectrom. 2021 Dec 1;32(12):2812-2820. doi: 10.1021/jasms.1c00245. Epub 2021 Nov 19. J Am Soc Mass Spectrom. 2021. PMID: 34797072 Free PMC article.
References
-
- Palha Joana A., Clinical Chemistry and Laboratory Medicine, 2002, 40, 1292. - PubMed
-
- Monaco HL, Biochimica et Biophysica Acta (BBA) - Protein Structure and Molecular Enzymology, 2000, 1482, 65–72. - PubMed
-
- Lai Z, Colón W and Kelly JW, Biochemistry, 1996, 35, 6470–6482. - PubMed
-
- Yoshiki S, Jeffery WK and Shu-ichi I, Current Pharmaceutical Design, 2008, 14, 3219–3230. - PubMed
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials

