ATP7A delivers copper to the lysyl oxidase family of enzymes and promotes tumorigenesis and metastasis
- PMID: 30890638
- PMCID: PMC6452744
- DOI: 10.1073/pnas.1817473116
ATP7A delivers copper to the lysyl oxidase family of enzymes and promotes tumorigenesis and metastasis
Abstract
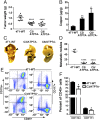
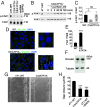
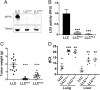
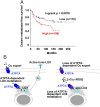
Lysyl oxidase (LOX) and LOX-like (LOXL) proteins are copper-dependent metalloenzymes with well-documented roles in tumor metastasis and fibrotic diseases. The mechanism by which copper is delivered to these enzymes is poorly understood. In this study, we demonstrate that the copper transporter ATP7A is necessary for the activity of LOX and LOXL enzymes. Silencing of ATP7A inhibited LOX activity in the 4T1 mammary carcinoma cell line, resulting in a loss of LOX-dependent mechanisms of metastasis, including the phosphorylation of focal adhesion kinase and myeloid cell recruitment to the lungs, in an orthotopic mouse model of breast cancer. ATP7A silencing was also found to attenuate LOX activity and metastasis of Lewis lung carcinoma cells in mice. Meta-analysis of breast cancer patients found that high ATP7A expression was significantly correlated with reduced survival. Taken together, these results identify ATP7A as a therapeutic target for blocking LOX- and LOXL-dependent malignancies.
Keywords: breast cancer; copper; lung cancer; lysyl oxidase; metastasis.
Conflict of interest statement
The authors declare no conflict of interest.
Figures

References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

