Sweet Cherry Fruit: Ideal Osmometers?
- PMID: 30891049
- PMCID: PMC6411844
- DOI: 10.3389/fpls.2019.00164
Sweet Cherry Fruit: Ideal Osmometers?
Abstract
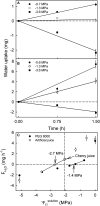
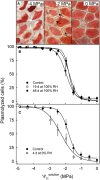
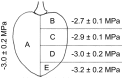
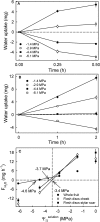
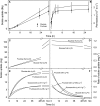
Osmotic water uptake through the skin is an important factor in rain cracking of sweet cherries. The objective was to establish whether a sweet cherry behaves like an ideal osmometer, where: (1) water uptake rates are negatively related to fruit osmotic potential, (2) a change in osmotic potential of the incubation solution results in a proportional change in water uptake rate, (3) the osmotic potential of the incubation solution yielding zero water uptake is numerically equal to the fruit water potential (in the absence of significant fruit turgor), and (4) the fruits' cuticular membrane is permeable only to water. The fruits' average osmotic potential and the rate of water uptake were related only weakly. Surprisingly, incubating a fruit in (a) the expressed juice from fruit of the same batch or (b) an isotonic artificial juice composed of the five major osmolytes of expressed juice or (c) an isotonic glucose solution-all resulted in significant water uptake. Decreasing the osmotic potential of the incubation solution decreased the rate of water uptake, while decreasing it still further resulted in water loss to the incubation solution. Throughout fruit development, the "apparent" fruit water potential was always more negative than the fruits' measured average osmotic potential. Plasmolysis of epidermal cells indicates the skin's osmotic potential was less negative than that of the flesh. When excised flesh discs were incubated in a concentration series of glucose solutions, the apparent water potential of the discs matched the osmotic potential of the expressed juice. Significant penetration of 14C-glucose and 14C-fructose occurred through excised fruit skins. These results indicate a sweet cherry is not an ideal osmometer. This is due in part to the cuticular membrane having a reflection coefficient for glucose and fructose less than unity. As a consequence, glucose and fructose were taken up by the fruit from the incubation solution. Furthermore, the osmotic potential of the expressed fruit juice is not uniform. The osmotic potential of juice taken from the stylar scar region is more negative than that from the pedicel region and that from the flesh more negative than that from the skin.
Keywords: cuticle; osmotic potential; prunus avium; reflection coefficient; water potential; water uptake.
Figures

References
-
- Becker T., Knoche M. (2011). Water movement through the surfaces of the grape berry and its stem. Am. J. Enol. Viticul. 62, 340–350. 10.5344/ajev.2011.10056 - DOI
-
- Beyer M., Peschel S., Knoche M., Knörgen M. (2002). Studies on water transport through the sweet cherry fruit surface: IV. Regions of preferential uptake. HortScience 37, 637–641. 10.21273/HORTSCI.37.4.637 - DOI
-
- Brüggenwirth M., Knoche M. (2016). Mechanical properties of skins of sweet cherry fruit of differing susceptibilities to cracking. J. Am. Soc. Horticult. Sci. 141, 162–168. 10.21273/JASHS.141.2.162 - DOI
-
- Bukovac M. J., Petracek P. D. (1993). Characterizing pesticide and surfactant penetration with isolated plant cuticles. Pesticide Sci. 37, 179–194. 10.1002/ps.2780370212 - DOI
-
- Christensen J. V. (1972). Cracking in cherries IV. determination of cracking susceptibility. Acta Agri. Scand. 22, 153–162. 10.1080/00015127209433474 - DOI
LinkOut - more resources
Full Text Sources
Research Materials

