Rapid pathology of lumpectomy margins with open-top light-sheet (OTLS) microscopy
- PMID: 30891344
- PMCID: PMC6420271
- DOI: 10.1364/BOE.10.001257
Rapid pathology of lumpectomy margins with open-top light-sheet (OTLS) microscopy
Abstract
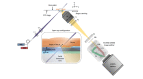
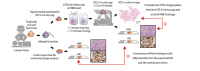
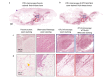
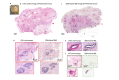
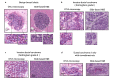
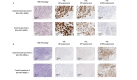
Open-top light-sheet microscopy is a technique that can potentially enable rapid ex vivo inspection of large tissue surfaces and volumes. Here, we have optimized an open-top light-sheet (OTLS) microscope and image-processing workflow for the comprehensive examination of surgical margin surfaces, and have also developed a novel fluorescent analog of H&E staining that is robust for staining fresh unfixed tissues. Our tissue-staining method can be achieved within 2.5 minutes followed by OTLS microscopy of lumpectomy surfaces at a rate of up to 1.5 cm2/minute. An image atlas is presented to show that OTLS image quality surpasses that of intraoperative frozen sectioning and can approximate that of gold-standard H&E histology of formalin-fixed paraffin-embedded (FFPE) tissues. Qualitative evidence indicates that these intraoperative methods do not interfere with downstream post-operative H&E histology and immunohistochemistry. These results should facilitate the translation of OTLS microscopy for intraoperative guidance of lumpectomy and other surgical oncology procedures.
Conflict of interest statement
A.K.G., N.P.R., and J.T.C.L. are cofounders and shareholders of LightSpeed Microscopy Inc.
Figures

References
-
- Fisher B., Anderson S., Bryant J., Margolese R. G., Deutsch M., Fisher E. R., Jeong J. H., Wolmark N., “Twenty-year follow-up of a randomized trial comparing total mastectomy, lumpectomy, and lumpectomy plus irradiation for the treatment of invasive breast cancer,” N. Engl. J. Med. 347(16), 1233–1241 (2002). 10.1056/NEJMoa022152 - DOI - PubMed
-
- Jacobson J. A., Danforth D. N., Cowan K. H., d’Angelo T., Steinberg S. M., Pierce L., Lippman M. E., Lichter A. S., Glatstein E., Okunieff P., “Ten-year results of a comparison of conservation with mastectomy in the treatment of stage I and II breast cancer,” N. Engl. J. Med. 332(14), 907–911 (1995). 10.1056/NEJM199504063321402 - DOI - PubMed
-
- Gage I., Schnitt S. J., Nixon A. J., Silver B., Recht A., Troyan S. L., Eberlein T., Love S. M., Gelman R., Harris J. R., Connolly J. L., “Pathologic margin involvement and the risk of recurrence in patients treated with breast-conserving therapy,” Cancer 78(9), 1921–1928 (1996). 10.1002/(SICI)1097-0142(19961101)78:9<1921::AID-CNCR12>3.0.CO;2-# - DOI - PubMed
Grants and funding
LinkOut - more resources
Full Text Sources
