Comparative histopathologic and viral immunohistochemical studies on CeMV infection among Western Mediterranean, Northeast-Central, and Southwestern Atlantic cetaceans
- PMID: 30893365
- PMCID: PMC6426187
- DOI: 10.1371/journal.pone.0213363
Comparative histopathologic and viral immunohistochemical studies on CeMV infection among Western Mediterranean, Northeast-Central, and Southwestern Atlantic cetaceans
Abstract
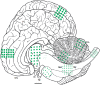
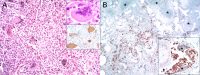
Cetacean morbillivirus (CeMV) is a major natural cause of morbidity and mortality in cetaceans worldwide and results in epidemic and endemic fatalities. The pathogenesis of CeMV has not been fully elucidated, and questions remain regarding tissue tropism and the mechanisms of immunosuppression. We compared the histopathologic and viral immunohistochemical features in molecularly confirmed CeMV-infected Guiana dolphins (Sotalia guianensis) from the Southwestern Atlantic (Brazil) and striped dolphins (Stenella coeruleoalba) and bottlenose dolphins (Tursiops truncatus) from the Northeast-Central Atlantic (Canary Islands, Spain) and the Western Mediterranean Sea (Italy). Major emphasis was placed on the central nervous system (CNS), including neuroanatomical distribution of lesions, and the lymphoid system and lung were also examined. Eleven Guiana dolphins, 13 striped dolphins, and 3 bottlenose dolphins were selected by defined criteria. CeMV infections showed a remarkable neurotropism in striped dolphins and bottlenose dolphins, while this was a rare feature in CeMV-infected Guiana dolphins. Neuroanatomical distribution of lesions in dolphins stranded in the Canary Islands revealed a consistent involvement of the cerebrum, thalamus, and cerebellum, followed by caudal brainstem and spinal cord. In most cases, Guiana dolphins had more severe lung lesions. The lymphoid system was involved in all three species, with consistent lymphoid depletion. Multinucleate giant cells/syncytia and characteristic viral inclusion bodies were variably observed in these organs. Overall, there was widespread lymphohistiocytic, epithelial, and neuronal/neuroglial viral antigen immunolabeling with some individual, host species, and CeMV strain differences. Preexisting and opportunistic infections were common, particularly endoparasitism, followed by bacterial, fungal, and viral infections. These results contribute to understanding CeMV infections in susceptible cetacean hosts in relation to factors such as CeMV strains and geographic locations, thereby establishing the basis for future neuro- and immunopathological comparative investigations.
Conflict of interest statement
The authors have declared that no competing interests exist.
Figures


References
-
- Domingo M, Vilafranca M, Visa J, Prats N, Trudgett A, Visser I. Evidence for chronic morbillivirus infection in the Mediterranean striped dolphin (Stenella coeruleoalba). Vet Microbiol. 1995; 44 (2–4): 229–239. - PubMed
Publication types
MeSH terms
Supplementary concepts
LinkOut - more resources
Full Text Sources

