Obesity and Fat Metabolism in Human Immunodeficiency Virus-Infected Individuals: Immunopathogenic Mechanisms and Clinical Implications
- PMID: 30893434
- PMCID: PMC6941618
- DOI: 10.1093/infdis/jiz118
Obesity and Fat Metabolism in Human Immunodeficiency Virus-Infected Individuals: Immunopathogenic Mechanisms and Clinical Implications
Abstract
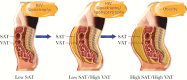
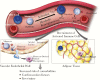
Metabolic complications relating to complex effects of viral and immune-mediated mechanisms are now a focus of clinical care among persons living with human immunodeficiency virus (PLHIV), and obesity is emerging as a critical problem. To address knowledge gaps, the US National Institutes of Health sponsored a symposium in May 2018 entitled "Obesity and Fat Metabolism in HIV-infected Individuals." Mechanisms relating to adipose dysfunction and fibrosis, immune function, inflammation, and gastrointestinal integrity were highlighted as contributors to obesity among PLHIV. Fibrotic subcutaneous adipose tissue is metabolically dysfunctional and loses its capacity to expand, leading to fat redistribution, including visceral obesity and ectopic fat accumulation, promoting insulin resistance. Viral proteins, including viral protein R and negative regulatory factor, have effects on adipogenic pathways and cellular metabolism in resident macrophages and T cells. HIV also affects immune cell trafficking into the adipose compartments, with effects on adipogenesis, lipolysis, and ectopic fat accumulation. Key cellular metabolic functions are likely to be affected in PLHIV by gut-derived cytokines and altered microbiota. There are limited strategies to reduce obesity specifically in PLHIV. Enhancing our understanding of critical pathogenic mechanisms will enable the development of novel therapeutics that may normalize adipose tissue function and distribution, reduce inflammation, and improve insulin sensitivity in PLHIV.
Keywords: HIV; inflammation; metabolism; obesity; viral proteins.
Published by Oxford University Press for the Infectious Diseases Society of America 2019.
Figures



References
-
- Achhra AC, Sabin C, Ryom L, et al. Body mass index and the risk of serious non-aids events and all-cause mortality in treated HIV-positive individuals: D: A: D cohort analysis. J Acquir Immune Defic Syndr 2018; 78:579–88. - PubMed
-
- Alexopoulos N, Katritsis D, Raggi P. Visceral adipose tissue as a source of inflammation and promoter of atherosclerosis. Atherosclerosis 2014; 233:104–12. - PubMed

