In situ 10-cell RNA sequencing in tissue and tumor biopsy samples
- PMID: 30894605
- PMCID: PMC6426952
- DOI: 10.1038/s41598-019-41235-9
In situ 10-cell RNA sequencing in tissue and tumor biopsy samples
Abstract
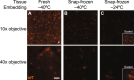
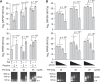
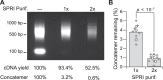
Single-cell transcriptomic methods classify new and existing cell types very effectively, but alternative approaches are needed to quantify the individual regulatory states of cells in their native tissue context. We combined the tissue preservation and single-cell resolution of laser capture with an improved preamplification procedure enabling RNA sequencing of 10 microdissected cells. This in situ 10-cell RNA sequencing (10cRNA-seq) can exploit fluorescent reporters of cell type in genetically engineered mice and is compatible with freshly cryoembedded clinical biopsies from patients. Through recombinant RNA spike-ins, we estimate dropout-free technical reliability as low as ~250 copies and a 50% detection sensitivity of ~45 copies per 10-cell reaction. By using small pools of microdissected cells, 10cRNA-seq improves technical per-cell reliability and sensitivity beyond existing approaches for single-cell RNA sequencing (scRNA-seq). Detection of low-abundance transcripts by 10cRNA-seq is comparable to random 10-cell groups of scRNA-seq data, suggesting no loss of gene recovery when cells are isolated in situ. Combined with existing approaches to deconvolve small pools of cells, 10cRNA-seq offers a reliable, unbiased, and sensitive way to measure cell-state heterogeneity in tissues and tumors.
Conflict of interest statement
The authors declare no competing interests.
Figures
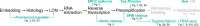



Similar articles
-
Single-Cell Transcriptomics of Immune Cells: Cell Isolation and cDNA Library Generation for scRNA-Seq.Methods Mol Biol. 2020;2184:1-18. doi: 10.1007/978-1-0716-0802-9_1. Methods Mol Biol. 2020. PMID: 32808214
-
Single-Cell Capture, RNA-seq, and Transcriptome Analysis from the Neural Retina.Methods Mol Biol. 2020;2092:159-186. doi: 10.1007/978-1-0716-0175-4_12. Methods Mol Biol. 2020. PMID: 31786788
-
CONICS integrates scRNA-seq with DNA sequencing to map gene expression to tumor sub-clones.Bioinformatics. 2018 Sep 15;34(18):3217-3219. doi: 10.1093/bioinformatics/bty316. Bioinformatics. 2018. PMID: 29897414 Free PMC article.
-
The promise of single-cell RNA sequencing for kidney disease investigation.Kidney Int. 2017 Dec;92(6):1334-1342. doi: 10.1016/j.kint.2017.06.033. Epub 2017 Oct 12. Kidney Int. 2017. PMID: 28893418 Free PMC article. Review.
-
Identifying cell types to interpret scRNA-seq data: how, why and more possibilities.Brief Funct Genomics. 2020 Jul 29;19(4):286-291. doi: 10.1093/bfgp/elaa003. Brief Funct Genomics. 2020. PMID: 32232401 Review.
Cited by
-
Applications of single-cell sequencing in cancer research: progress and perspectives.J Hematol Oncol. 2021 Jun 9;14(1):91. doi: 10.1186/s13045-021-01105-2. J Hematol Oncol. 2021. PMID: 34108022 Free PMC article. Review.
-
Sporadic activation of an oxidative stress-dependent NRF2-p53 signaling network in breast epithelial spheroids and premalignancies.Sci Signal. 2020 Apr 14;13(627):eaba4200. doi: 10.1126/scisignal.aba4200. Sci Signal. 2020. PMID: 32291314 Free PMC article.
-
Use of Single-Cell -Omic Technologies to Study the Gastrointestinal Tract and Diseases, From Single Cell Identities to Patient Features.Gastroenterology. 2020 Aug;159(2):453-466.e1. doi: 10.1053/j.gastro.2020.04.073. Epub 2020 May 14. Gastroenterology. 2020. PMID: 32417404 Free PMC article. Review.
-
Modeling the complete kinetics of coxsackievirus B3 reveals human determinants of host-cell feedback.Cell Syst. 2021 Apr 21;12(4):304-323.e13. doi: 10.1016/j.cels.2021.02.004. Epub 2021 Mar 18. Cell Syst. 2021. PMID: 33740397 Free PMC article.
-
Olig2-astrocytes express neutral amino acid transporter SLC7A10 (Asc-1) in the adult brain.Mol Brain. 2021 Nov 8;14(1):163. doi: 10.1186/s13041-021-00874-8. Mol Brain. 2021. PMID: 34749773 Free PMC article.
References
-
- Fidler IJ. Tumor heterogeneity and the biology of cancer invasion and metastasis. Cancer Res. 1978;38:2651–2660. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

