Low levels of SIV-specific CD8+ T cells in germinal centers characterizes acute SIV infection
- PMID: 30897187
- PMCID: PMC6445460
- DOI: 10.1371/journal.ppat.1007311
Low levels of SIV-specific CD8+ T cells in germinal centers characterizes acute SIV infection
Abstract
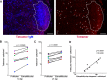
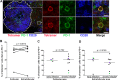
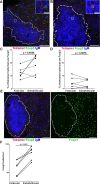
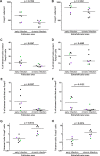
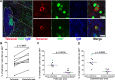
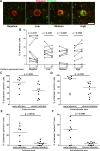
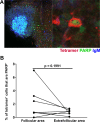
CD8+ T cells play an important role in controlling of HIV and SIV infections. However, these cells are largely excluded from B cell follicles where HIV and SIV producing cells concentrate during chronic infection. It is not known, however, if antigen-specific CD8+ T cells are excluded gradually as pathogenesis progresses from early to chronic phase, or this phenomenon occurs from the beginning infection. In this study we determined that SIV-specific CD8+ T cells were largely excluded from follicles during early infection, we also found that within follicles, they were entirely absent in 60% of the germinal centers (GCs) examined. Furthermore, levels of SIV-specific CD8+ T cells in follicular but not extrafollicular areas significantly correlated inversely with levels of viral RNA+ cells. In addition, subsets of follicular SIV-specific CD8+ T cells were activated and proliferating and expressed the cytolytic protein perforin. These studies suggest that a paucity of SIV-specific CD8+ T cells in follicles and complete absence within GCs during early infection may set the stage for the establishment of persistent chronic infection.
Conflict of interest statement
The authors have declared that no competing interests exist.
Figures


Similar articles
-
Simian Immunodeficiency Virus-Producing Cells in Follicles Are Partially Suppressed by CD8+ Cells In Vivo.J Virol. 2016 Nov 28;90(24):11168-11180. doi: 10.1128/JVI.01332-16. Print 2016 Dec 15. J Virol. 2016. PMID: 27707919 Free PMC article.
-
Spatial alterations between CD4(+) T follicular helper, B, and CD8(+) T cells during simian immunodeficiency virus infection: T/B cell homeostasis, activation, and potential mechanism for viral escape.J Immunol. 2012 Apr 1;188(7):3247-56. doi: 10.4049/jimmunol.1103138. Epub 2012 Mar 2. J Immunol. 2012. PMID: 22387550 Free PMC article.
-
CAR/CXCR5-T cell immunotherapy is safe and potentially efficacious in promoting sustained remission of SIV infection.PLoS Pathog. 2022 Feb 7;18(2):e1009831. doi: 10.1371/journal.ppat.1009831. eCollection 2022 Feb. PLoS Pathog. 2022. PMID: 35130312 Free PMC article.
-
Tfh1 Cells in Germinal Centers During Chronic HIV/SIV Infection.Front Immunol. 2018 Jun 6;9:1272. doi: 10.3389/fimmu.2018.01272. eCollection 2018. Front Immunol. 2018. PMID: 29928280 Free PMC article. Review.
-
Mechanisms of CD8+ T cell-mediated suppression of HIV/SIV replication.Eur J Immunol. 2018 Jun;48(6):898-914. doi: 10.1002/eji.201747172. Epub 2018 Mar 26. Eur J Immunol. 2018. PMID: 29427516 Free PMC article. Review.
Cited by
-
Development of an anti-CAR antibody response in SIV-infected rhesus macaques treated with CD4-MBL CAR/CXCR5 T cells.Front Immunol. 2022 Dec 13;13:1032537. doi: 10.3389/fimmu.2022.1032537. eCollection 2022. Front Immunol. 2022. PMID: 36582226 Free PMC article.
-
Evolution and Diversity of Immune Responses during Acute HIV Infection.Immunity. 2020 Nov 17;53(5):908-924. doi: 10.1016/j.immuni.2020.10.015. Immunity. 2020. PMID: 33207216 Free PMC article. Review.
-
Cytolytic CD8+ T cells infiltrate germinal centers to limit ongoing HIV replication in spontaneous controller lymph nodes.Sci Immunol. 2023 May 19;8(83):eade5872. doi: 10.1126/sciimmunol.ade5872. Epub 2023 May 19. Sci Immunol. 2023. PMID: 37205767 Free PMC article.
-
Detection of Antigen-Specific T Cells Using In Situ MHC Tetramer Staining.Int J Mol Sci. 2019 Oct 18;20(20):5165. doi: 10.3390/ijms20205165. Int J Mol Sci. 2019. PMID: 31635220 Free PMC article. Review.
-
The impact of HIV infection on the frequencies, function, spatial localization and heterogeneity of T follicular regulatory cells (TFRs) within human lymph nodes.BMC Immunol. 2022 Jul 1;23(1):34. doi: 10.1186/s12865-022-00508-1. BMC Immunol. 2022. PMID: 35778692 Free PMC article.
References
-
- Connick E, Folkvord JM, Lind KT, Rakasz EG, Miles B, Wilson NA, et al. Compartmentalization of simian immunodeficiency virus replication within secondary lymphoid tissues of rhesus macaques is linked to disease stage and inversely related to localization of virus-specific CTL. J Immunol. American Association of Immunologists; 2014;193: 5613–25. 10.4049/jimmunol.1401161 - DOI - PMC - PubMed
-
- Hufert FT, van Lunzen J, Janossy G, Bertram S, Schmitz J, Haller O, et al. Germinal centre CD4+ T cells are an important site of HIV replication in vivo. AIDS. 1997;11 Available: http://journals.lww.com/aidsonline/Fulltext/1997/07000/Germinal_centre_C... - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

