Anti-Oxidative and Anti-Aging Activities of Porcine By-Product Collagen Hydrolysates Produced by Commercial Proteases: Effect of Hydrolysis and Ultrafiltration
- PMID: 30897764
- PMCID: PMC6472052
- DOI: 10.3390/molecules24061104
Anti-Oxidative and Anti-Aging Activities of Porcine By-Product Collagen Hydrolysates Produced by Commercial Proteases: Effect of Hydrolysis and Ultrafiltration
Abstract
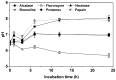
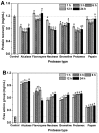
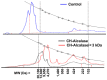
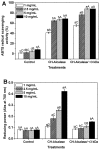
To investigate methods for improving the processing of porcine waste, porcine skin was hydrolyzed using different commercially available proteases (Alcalase, Flavorzyme, Neutrase, Bromeline, Protamex, and Papain) under several optimal conditions. Following enzymatic hydrolysis, the collagen hydrolysates (CHs) were fractionated by molecular weight (3 kDa) via membrane ultrafiltration. The CHs were analyzed for physical properties (pH, protein recovery, free amino group content, molecular weight distribution, and amino composition) as well as for functional properties (antioxidant activities and anti-aging activities). Among the CHs, CHs hydrolyzed by Alcalase (CH-Alcalase) exhibited the highest degree of hydrolysis compared to other CHs. Both "CH-Alcalase" and "CH-Alcalase < 3 kDa" fractions showed a considerably high antioxidant activity and collagenase inhibition activity. Therefore, resulting bioactives have potential for development as antioxidants and anti-aging ingredients in the food, cosmetics, and pharmaceuticals, from animal by-products.
Keywords: anti-aging; antioxidant; collagen hydrolysate; commercial proteases; hydrolysis.
Conflict of interest statement
The authors declare no conflict of interest.
Figures

References
-
- Je J.-Y., Qian Z.-J., Byun H.-G., Kim S.-K. Purification and characterization of an antioxidant peptide obtained from tuna backbone protein by enzymatic hydrolysis. Process Biochem. 2007;42:840–846. doi: 10.1016/j.procbio.2007.02.006. - DOI
-
- Agyei D., Ongkudon C.M., Wei C.Y., Chan A.S., Danquah M.K. Bioprocess challenges to the isolation and purification of bioactive peptides. Chem. Eng. Res. Des. 2016;98:244–256. doi: 10.1016/j.fbp.2016.02.003. - DOI
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

