FOXK2 Transcription Factor and Its Emerging Roles in Cancer
- PMID: 30897782
- PMCID: PMC6468357
- DOI: 10.3390/cancers11030393
FOXK2 Transcription Factor and Its Emerging Roles in Cancer
Abstract
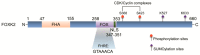
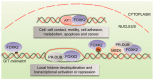
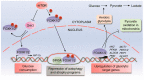
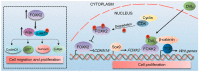
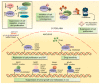
Forkhead box (FOX) transcription factors compose a large family of regulators of key biological processes within a cell. FOXK2 is a member of FOX family, whose biological functions remain relatively unexplored, despite its description in the early nineties. More recently, growing evidence has been pointing towards a role of FOXK2 in cancer, which is likely to be context-dependent and tumour-specific. Here, we provide an overview of important aspects concerning the mechanisms of regulation of FOXK2 expression and function, as well as its complex interactions at the chromatin level, which orchestrate how it differentially regulates the expression of gene targets in pathophysiology. Particularly, we explore the emerging functions of FOXK2 as a regulator of a broad range of cancer features, such as cell proliferation and survival, DNA damage, metabolism, migration, invasion and metastasis. Finally, we discuss the prognostic value of assessing FOXK2 expression in cancer patients and how it can be potentially targeted for future anticancer interventions.
Keywords: FOXK2 transcription factor; cancer; forkhead box; transcriptional activity.
Conflict of interest statement
The authors declare no conflict of interest.
Figures
Similar articles
-
FOXK2 transcription factor and its roles in tumorigenesis (Review).Oncol Lett. 2022 Nov 3;24(6):461. doi: 10.3892/ol.2022.13581. eCollection 2022 Dec. Oncol Lett. 2022. PMID: 36380871 Free PMC article. Review.
-
Forkhead box K2 promotes human colorectal cancer metastasis by upregulating ZEB1 and EGFR.Theranostics. 2019 May 31;9(13):3879-3902. doi: 10.7150/thno.31716. eCollection 2019. Theranostics. 2019. PMID: 31281520 Free PMC article.
-
Regulation and roles of FOXK2 in cancer.Front Oncol. 2022 Sep 12;12:967625. doi: 10.3389/fonc.2022.967625. eCollection 2022. Front Oncol. 2022. PMID: 36172141 Free PMC article. Review.
-
Downregulation of FOXK2 is associated with poor prognosis in patients with gastric cancer.Mol Med Rep. 2018 Nov;18(5):4356-4364. doi: 10.3892/mmr.2018.9466. Epub 2018 Sep 7. Mol Med Rep. 2018. PMID: 30221666 Free PMC article.
-
Forkhead box K2 inhibits the proliferation, migration, and invasion of human glioma cells and predicts a favorable prognosis.Onco Targets Ther. 2018 Feb 27;11:1067-1075. doi: 10.2147/OTT.S157126. eCollection 2018. Onco Targets Ther. 2018. PMID: 29520156 Free PMC article.
Cited by
-
Targeting Transcription Factors in Cancer: From "Undruggable" to "Druggable".Methods Mol Biol. 2023;2594:107-131. doi: 10.1007/978-1-0716-2815-7_9. Methods Mol Biol. 2023. PMID: 36264492 Free PMC article.
-
Integrating the Epigenome and Transcriptome of Hepatocellular Carcinoma to Identify Systematic Enhancer Aberrations and Establish an Aberrant Enhancer-Related Prognostic Signature.Front Cell Dev Biol. 2022 Mar 1;10:827657. doi: 10.3389/fcell.2022.827657. eCollection 2022. Front Cell Dev Biol. 2022. PMID: 35300417 Free PMC article.
-
PR-DUB maintains the expression of critical genes through FOXK1/2- and ASXL1/2/3-dependent recruitment to chromatin and H2AK119ub1 deubiquitination.Genome Res. 2020 Aug;30(8):1119-1130. doi: 10.1101/gr.261016.120. Epub 2020 Aug 3. Genome Res. 2020. PMID: 32747411 Free PMC article.
-
Systematic Review and Meta-analysis of Peripheral Blood DNA Methylation Studies in Inflammatory Bowel Disease.J Crohns Colitis. 2023 Mar 18;17(2):185-198. doi: 10.1093/ecco-jcc/jjac119. J Crohns Colitis. 2023. PMID: 35998097 Free PMC article.
-
Emerging roles of FOXK2 in cancers and metabolic disorders.Front Oncol. 2024 Apr 29;14:1376496. doi: 10.3389/fonc.2024.1376496. eCollection 2024. Front Oncol. 2024. PMID: 38741782 Free PMC article. Review.
References
-
- Kaestner K.H., Knochel W., Martinez D.E. Unified nomenclature for the winged helix/forkhead transcription factors. Genes Dev. 2000;14:142–146. - PubMed
Publication types
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

