Preparation of Sesquiterpene Lactone Derivatives: Cytotoxic Activity and Selectivity of Action
- PMID: 30897836
- PMCID: PMC6471591
- DOI: 10.3390/molecules24061113
Preparation of Sesquiterpene Lactone Derivatives: Cytotoxic Activity and Selectivity of Action
Abstract
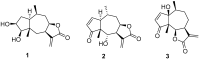
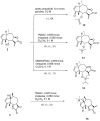
Cancer is one of the most important causes of death worldwide. Solid tumors represent the great majority of cancers (>90%) and the chemotherapeutic agents used for their treatment are still characterized by variable efficacy and toxicity. Sesquiterpene lactones are a group of naturally occurring compounds that have displayed a diverse range of biological activities including cytotoxic activity. A series of oxygenated and oxy-nitrogenated derivatives (4⁻15) from the sesquiterpene lactones cumanin (1), helenalin (2), and hymenin (3) were synthesized. The silylated derivatives of helenalin, compounds 13 and 14, were found to be the most active against tumor cell lines, with GI50 values ranging from 0.15 to 0.59 μM. The ditriazolyl cumanin derivative (11) proved to be more active and selective than cumanin in the tested breast, cervix, lung, and colon tumor cell lines. This compound was the least toxic against splenocytes (CC50 = 524.1 µM) and exhibited the greatest selectivity on tumor cell lines. This compound showed a GI50 of 2.3 µM and a SI of 227.9 on WiDr human colon tumor cell lines. Thus, compound 11 can be considered for further studies and is a candidate for the development of new antitumor agents.
Keywords: Asteraceae; antiproliferative activity; cumanin; helenalin; hymenin; sesquiterpene lactones.
Conflict of interest statement
The authors declare no conflict of interest.
Figures



References
-
- Lahlou M. The success of natural products in drug discovery. Pharmacol. Pharm. 2013;4:17–31. doi: 10.4236/pp.2013.43A003. - DOI
-
- Sülsen V.P., Frank F.M., Cazorla S.I., Anesini C.A., Malchiodi E.L., Freixa B., Martino V.S. Trypanocidal and leishmanicidal activities of sesquiterpene lactones from Ambrosia tenuifolia Sprengel (Asteraceae) Antimicrob. Agent. Chemother. 2008;52:2415–2419. doi: 10.1128/AAC.01630-07. - DOI - PMC - PubMed
-
- Sülsen V.P., Cazorla S.I., Frank F.M., Laurella L.C., Muschietti L.V., Catalan C.A., Malchiodi E.L. Natural terpenoids from Ambrosia species are active in vitro and in vivo against human pathogenic trypanosomatids. PLoS Negl. Trop. Dis. 2013;7:e2494. doi: 10.1371/journal.pntd.0002494. - DOI - PMC - PubMed
MeSH terms
Substances
Grants and funding
- PIP-090/Consejo Nacional de Investigaciones Científicas y Técnicas
- PROICO 2-2516/Universidad Nacional de San Luis
- PIP 11220150100158CO/Consejo Nacional de Investigaciones Científicas y Técnicas
- PICT 2015-3531/Agencia Nacional de Promoción Científica y Tecnológica
- UBACYT 20020130200270/Universidad de Buenos Aires
LinkOut - more resources
Full Text Sources

